Inter-laboratory variability in cytomegalovirus DNA quantification: implications for standardization and clinical monitoring
- PMID: 40470970
- PMCID: PMC12239724
- DOI: 10.1128/jcm.01911-24
Inter-laboratory variability in cytomegalovirus DNA quantification: implications for standardization and clinical monitoring
Abstract
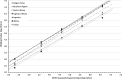
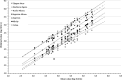
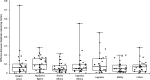
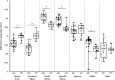
Cytomegalovirus (CMV) infection monitoring is a key element in the management of immunocompromised patients. CMV DNA quantification in plasma or whole blood is the best indicator for clinicians to adjust immunosuppressive or antiviral therapies. Despite the availability of internationally standardized material, the commutability of CMV quantification results across laboratories remains inadequate. To assess inter-laboratory variability in CMV DNA quantification, we conducted a blinded study in seven independent laboratories. Each participant received a panel of 92 specimens for CMV quantification using their routinely used standard platform. While quantifications were highly correlated and reproducible, large discrepancies were observed with differences up to 1.45 log10 IU/mL between techniques for identical specimens. However, quantification scattering was lower for the World Health Organization (WHO) international standard or a commercially tested control (interquartile range = 0.129) than for clinical specimens (0.469; P = 0.0142). Blind quantification of the WHO or the commercial standard indicated that all techniques, except for fully integrated platforms, did not align well with the expected values, and most platforms tended to quantify specimens and standards differently. Recalibration of all platforms against the same standard improved the spread of results, but differences of up to 1.19 log10 IU/mL remained for the same specimens. Achieving commutability in CMV quantification remains an elusive goal. Efforts should focus on improving both the assay calibrators and the run controls, which currently do not appear to simulate the unique characteristics of circulating CMV in patients. Until this is resolved, each transplanted patient should be consistently monitored by the same laboratory on the same platform.IMPORTANCEOur conclusions support previous work on this topic describing the diversity of circulating cytomegalovirus (CMV) DNA forms and the difficulties in standardizing CMV viral load (VL) measurement. The inter-assay reproducibility of CMV VL measurement is primarily influenced by the extraction procedure and the amplicon size generated by the technique. Viral standards generated from cell culture supernatant do not reflect circulating CMV forms from patient samples. We highlight the need to develop a new international standard that better reflects the circulating forms of CMV and demonstrate the risk of tracking a patient's CMV VL in different laboratories.
Keywords: calibration; extraction; fragmentation; immunosuppression; viral load monitoring.
Conflict of interest statement
Bio-Rad provided financial support for the constitution of the panel and covered the cost of CMV viral load determination to each laboratory.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

