A novel peptide mimetic, brilacidin, for combating multidrug-resistant Neisseria gonorrhoeae
- PMID: 40471977
- PMCID: PMC12140270
- DOI: 10.1371/journal.pone.0325722
A novel peptide mimetic, brilacidin, for combating multidrug-resistant Neisseria gonorrhoeae
Abstract
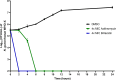
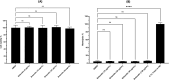
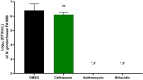
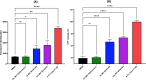
Neisseria gonorrhoeae is classified by the Centers for Disease Control and Prevention as an urgent public health threat due to rising infections and rapid resistance development. N. gonorrhoeae has developed resistance to nearly all FDA-approved drugs, with ceftriaxone being the only remaining effective treatment for gonococcal infections. Alarmingly, ceftriaxone-resistant N. gonorrhoeae strains were isolated worldwide, raising the potential of untreatable gonorrhea in the near future. Hence, the critical need to develop new anti-N. gonorrhoeae therapeutics cannot be overemphasized. In this study, we identified the peptide mimetic brilacidin as an effective anti-gonococcal agent. Brilacidin completed phase 2 clinical trials for treating skin infections, oral mucositis, and COVID-19. Herein, brilacidin displayed potent activity against a panel of 22 drug-resistant strains of N. gonorrhoeae, inhibiting 50% of the strains tested (MIC50) at the concentration of 4 µg/mL. The peptide exhibited rapid bactericidal activity, reducing N. gonorrhoeae high inoculum within two hours. Moreover, brilacidin was superior to the drug of choice, ceftriaxone, in eliminating the intracellular N. gonorrhoeae harbored within endocervical cells. Additionally, brilacidin showed high tolerability in mammalian cells and lacked hemolytic activity in human erythrocytes. Altogether, the results demonstrate that brilacidin is a promising anti-gonococcal agent that warrants further in-depth investigation.
Copyright: © 2025 Abdelsattar et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Cyclization increases bactericidal activity of arginine-rich cationic cell-penetrating peptide for Neisseria gonorrhoeae.Microbiol Spectr. 2024 Sep 3;12(9):e0099724. doi: 10.1128/spectrum.00997-24. Epub 2024 Aug 6. Microbiol Spectr. 2024. PMID: 39105587 Free PMC article.
-
Investigation of auranofin and gold-containing analogues antibacterial activity against multidrug-resistant Neisseria gonorrhoeae.Sci Rep. 2020 Mar 27;10(1):5602. doi: 10.1038/s41598-020-62696-3. Sci Rep. 2020. PMID: 32221472 Free PMC article.
-
Neisseria gonorrhoeae Antimicrobial Susceptibility Surveillance - The Gonococcal Isolate Surveillance Project, 27 Sites, United States, 2014.MMWR Surveill Summ. 2016 Jul 15;65(7):1-19. doi: 10.15585/mmwr.ss6507a1. MMWR Surveill Summ. 2016. PMID: 27414503
-
Multiresistant Neisseria gonorrhoeae: a new threat in second decade of the XXI century.Med Microbiol Immunol. 2020 Apr;209(2):95-108. doi: 10.1007/s00430-019-00651-4. Epub 2019 Dec 4. Med Microbiol Immunol. 2020. PMID: 31802195 Free PMC article. Review.
-
Update on Treatment Options for Gonococcal Infections.Pharmacotherapy. 2015 Sep;35(9):856-68. doi: 10.1002/phar.1627. Epub 2015 Sep 6. Pharmacotherapy. 2015. PMID: 26343813 Review.
References
-
- Newman L, Rowley J, Vander Hoorn S, Wijesooriya NS, Unemo M, Low N, et al.. Global Estimates of the Prevalence and Incidence of Four Curable Sexually Transmitted Infections in 2012 Based on Systematic Review and Global Reporting. PLoS One. 2015;10(12):e0143304. doi: 10.1371/journal.pone.0143304 - DOI - PMC - PubMed
-
- Gonococcal Infections Among Adolescents and Adults - STI Treatment Guidelines. 5 Dec 2022. [cited 14 Aug 2024]. Available: https://www.cdc.gov/std/treatment-guidelines/gonorrhea-adults.htm
-
- Gonorrhoea: latest antimicrobial global surveillance results and guidance for vaccine development published. [cited 14 Apr 2025] Available: https://www.who.int/news/item/22-11-2021-gonorrhoea-antimicrobial-resist...
-
- Sexually transmitted infections (STIs). [cited 14 Apr 2025]. Available: https://www.who.int/news-room/fact-sheets/detail/sexually-transmitted-in...)
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

