Chalkophore-mediated respiratory oxidase flexibility controls M. tuberculosis virulence
- PMID: 40472191
- PMCID: PMC12140626
- DOI: 10.7554/eLife.105794
Chalkophore-mediated respiratory oxidase flexibility controls M. tuberculosis virulence
Abstract
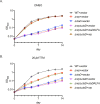
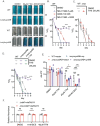
Oxidative phosphorylation has emerged as a critical therapeutic vulnerability of M. tuberculosis (Mtb). However, it is unknown how intracellular bacterial pathogens such as Mtb maintain respiration during infection despite the chemical effectors of host immunity. Mtb synthesizes diisonitrile lipopeptides that tightly chelate copper, but the role of these chalkophores in host-pathogen interactions is also unknown. We demonstrate that M. tuberculosis chalkophores maintain the function of the heme-copper bcc:aa3 respiratory supercomplex under copper limitation. Chalkophore deficiency impairs Mtb survival, respiration to oxygen, and ATP production under copper deprivation in culture, effects that are exacerbated by loss of the heme-dependent Cytochrome BD respiratory oxidase. Our genetic analyses indicate that the maintenance of respiration is the major cellular target of chalkophore-mediated copper acquisition. M. tuberculosis lacking chalkophore biosynthesis is attenuated in mice, a phenotype that is also severely exacerbated by loss of the CytBD respiratory oxidase. We find that the host immune pressure that attenuates chalkophore-deficient Mtb is independent of adaptive immunity and neutrophils. These data demonstrate that chalkophores counter host-inflicted copper deprivation and highlight a multilayered system by which M. tuberculosis maintains respiration during infection.
Keywords: Copper; M. tuberculosis; infectious disease; microbiology; nutrional immunity; oxidative phosphorylation; tuberculosis.
Plain language summary
When a bacterium known as Mycobacterium tuberculosis infects humans, it can lead to a disease called tuberculosis – one of the leading causes of death from an infectious disease worldwide. The bacteria hide within certain cells in the body so that they are less accessible to the host’s immune system. This approach is so successful that patients typically require long courses of antibiotics lasting many months to eliminate the bacteria. However, the emergence of drug-resistant strains of M. tuberculosis means that new methods to target the bacteria are urgently needed. One possible target for future therapies is a system known as diisonitrile lipopeptide chalkophores, which bind to copper and import the metal into the bacterium from its surroundings. Although certain proteins in bacteria require copper to work properly, the target of the copper acquired by the chalkophores in M. tuberculosis has remained unclear. Here, Buglino, Ozakman et al. used genetic and biochemical approaches to study why diisonitrile lipopeptide chalkophores collect copper in M. tuberculosis. The experiments showed that the chalkophores supply copper to proteins with a key role in respiration, the process by which cells make chemical energy needed for many cell activities. When there was a shortage of copper in their surrounding environment, mutant M. tuberculosis cells lacking the chalkophores were less able to produce chemical energy and more likely to die than healthy M. tuberculosis cells. This effect was more severe if the cells were also missing an enzyme that enables the bacteria to respire without copper. Further experiments in mice found that during a tuberculosis infection, the immune system targeted copper-containing proteins in its attempts to kill the bacteria. The mutant M. tuberculosis cells were less effective at infecting the mice, suggesting that chalkophores help the bacteria defend themselves against the host immune system. Taken together, these findings reveal that copper-containing proteins in M. tuberculosis are a major target of the immune system. In the future, increasing our understanding of these proteins and identifying drugs that interfere with their activities may lead to new, more effective therapies for tuberculosis.
© 2025, Buglino, Ozakman et al.
Conflict of interest statement
JB, YO, CH, AB, DT No competing interests declared, MG Declares equity and consulting fees from Vedanta biosciences and consulting fees from Fimbrion therapeutics
Figures







Update of
-
Chalkophore mediated respiratory oxidase flexibility controls M. tuberculosis virulence.bioRxiv [Preprint]. 2025 Mar 4:2024.04.12.589290. doi: 10.1101/2024.04.12.589290. bioRxiv. 2025. Update in: Elife. 2025 Jun 05;14:RP105794. doi: 10.7554/eLife.105794. PMID: 38645185 Free PMC article. Updated. Preprint.
References
-
- Andrews S. FastQC: a quality control tool for high throughput sequence data. GPL v3Bioinformatics Babraham. 2010 http://www.bioinformatics.babraham.ac.uk/projects/fastqc
-
- Behnsen J, Zhi H, Aron AT, Subramanian V, Santus W, Lee MH, Gerner RR, Petras D, Liu JZ, Green KD, Price SL, Camacho J, Hillman H, Tjokrosurjo J, Montaldo NP, Hoover EM, Treacy-Abarca S, Gilston BA, Skaar EP, Chazin WJ, Garneau-Tsodikova S, Lawrenz MB, Perry RD, Nuccio SP, Dorrestein PC, Raffatellu M. Siderophore-mediated zinc acquisition enhances enterobacterial colonization of the inflamed gut. Nature Communications. 2021;12:7016. doi: 10.1038/s41467-021-27297-2. - DOI - PMC - PubMed
-
- Beites T, O’Brien K, Tiwari D, Engelhart CA, Walters S, Andrews J, Yang HJ, Sutphen ML, Weiner DM, Dayao EK, Zimmerman M, Prideaux B, Desai PV, Masquelin T, Via LE, Dartois V, Boshoff HI, Barry CE, Ehrt S, Schnappinger D. Plasticity of the Mycobacterium tuberculosis respiratory chain and its impact on tuberculosis drug development. Nature Communications. 2019;10:4970. doi: 10.1038/s41467-019-12956-2. - DOI - PMC - PubMed
-
- Bhatt K, Machado H, Osório NS, Sousa J, Cardoso F, Magalhães C, Chen B, Chen M, Kim J, Singh A, Ferreira CM, Castro AG, Torrado E, Jacobs WR, Jr, Bhatt A, Saraiva M. A nonribosomal peptide synthase gene driving virulence in Mycobacterium tuberculosis. mSphere. 2018;3:e00352-18. doi: 10.1128/mSphere.00352-18. - DOI - PMC - PubMed
-
- Bobrov AG, Kirillina O, Fetherston JD, Miller MC, Burlison JA, Perry RD. The Yersinia pestis siderophore, yersiniabactin, and the ZnuABC system both contribute to zinc acquisition and the development of lethal septicaemic plague in mice. Molecular Microbiology. 2014;93:759–775. doi: 10.1111/mmi.12693. - DOI - PMC - PubMed
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

