A gatekeeper sympathetic control of lacrimal tear secretion and dry eye onset through the NA-Adra1a-Ucp2 pathway
- PMID: 40473608
- PMCID: PMC12141490
- DOI: 10.1038/s41467-025-60476-z
A gatekeeper sympathetic control of lacrimal tear secretion and dry eye onset through the NA-Adra1a-Ucp2 pathway
Abstract
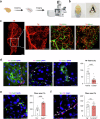
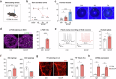
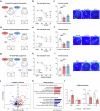
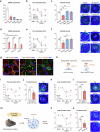
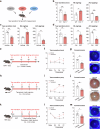
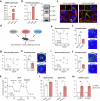
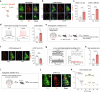
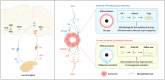
Tear secretion from the lacrimal gland is essential for maintaining ocular surface homeostasis, and its insufficiency causes aqueous-deficient dry eye. Unlike the well-established parasympathetic neuronal regulation, the role of sympathetic nervous system (SNS) in tear secretion remains controversial. Here, we demonstrate the intact sympathetic innervation in lacrimal gland and its activation under multiple dry eye stresses. Pharmacological, surgical, and genetic blockade of SNS increases tear secretion and alleviates dry eye signs. Mechanistically, SNS-driven noradrenaline (NA) release activates α1a-adrenergic receptor (Adra1a) in acinar and myoepithelial cells to regulate mitochondrial Ucp2 and tear secretion. Systemic and local delivery of Adra1a antagonists, including silodosin and tamsulosin, improves tear secretion and reduces corneal lesions in multiple dry eye mouse models. In addition, we identify the brain locus coeruleus as an upstream driver orchestrating sympathetic regulation of lacrimal secretion. Overall, these findings reveal a gatekeeper role of SNS in tear secretion and offer potential therapeutic strategies for dry eye disease.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: A Chinese patent (ZL202210497086.8) has been granted to Eye Institute of Shandong First Medical University (Shandong Eye Institute, Qingdao Eye Hospital of Shandong First Medical University), with inventors Lixin Xie, Qingjun Zhou, Mingli Qu, and Sai Zhang. It covers the use of sympathetic activation inhibitors and/or α1-adrenergic receptor antagonists for treating dry eye. A related PCT application (PCT/CN2022/113244) is under examination. All other authors declare no competing interests.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

