Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase gene fusion: report of two cases with different clinical presentation
- PMID: 40474715
- PMCID: PMC12142289
- DOI: 10.32074/1591-951X-975
Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase gene fusion: report of two cases with different clinical presentation
Abstract
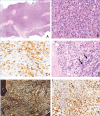
Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase gene fusions (M/LN-eo-TK) such as PDGFRA, PDGFRB, FGFR1, JAK2, FLT3 rearrangement and ETV6::ABL1 fusion include rare and heterogeneous clinical-pathological entities with some similarities, not always associated with peripheral eosinophilia. Accurate diagnosis and demonstration of the specific genetic substrate have important implications since target therapy is possibly available. Herein we report two cases showing different bone marrow features and clinical presentation. Recognition of eosinophilic granuloblasts prompted genetic analysis that showed PDGFRB (case 1) and PDGFRA (case 2) gene rearrangement. Diagnosis of M/LN-eo-TK may be challenging. Pathologists may be the first professionals to suspect the disorder and should be aware of the therapeutic implication. Accurate BOM marrow evaluation with a panel of immunohistochemical reactions, and specific molecular analyses are required for proper diagnosis.
Keywords: Hypereosinophilia; Myeloid/lymphoid neoplasms; PDGFRA; PDGFRB; TKI.
Copyright © 2025 Società Italiana di Anatomia Patologica e Citopatologia Diagnostica, Divisione Italiana della International Academy of Pathology.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
The Use of Optical Genome Mapping for the Detection of Tyrosine Kinase Gene Fusions in Myeloid/Lymphoid Neoplasms.J Cell Mol Med. 2025 Jun;29(12):e70640. doi: 10.1111/jcmm.70640. J Cell Mol Med. 2025. PMID: 40533930 Free PMC article.
-
Myeloid neoplasms with eosinophilia.Blood. 2017 Feb 9;129(6):704-714. doi: 10.1182/blood-2016-10-695973. Epub 2016 Dec 27. Blood. 2017. PMID: 28028030 Review.
-
Hematologic malignancies with PCM1-JAK2 gene fusion share characteristics with myeloid and lymphoid neoplasms with eosinophilia and abnormalities of PDGFRA, PDGFRB, and FGFR1.Ann Hematol. 2013 Jun;92(6):759-69. doi: 10.1007/s00277-013-1695-3. Epub 2013 Feb 12. Ann Hematol. 2013. PMID: 23400675
-
A novel TRIP11-FLT3 fusion in a patient with a myeloid/lymphoid neoplasm with eosinophilia.Cancer Genet. 2017 Oct;216-217:10-15. doi: 10.1016/j.cancergen.2017.05.001. Epub 2017 May 10. Cancer Genet. 2017. PMID: 29025582 Free PMC article.
-
Updates on eosinophilic disorders.Virchows Arch. 2023 Jan;482(1):85-97. doi: 10.1007/s00428-022-03402-8. Epub 2022 Sep 7. Virchows Arch. 2023. PMID: 36068374 Review.
References
-
- Shomali W, Gotlib J. World Health Organization-defined eosinophilic disorders: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97(1):129-148. https://doi.org/10.1002/ajh.26352 10.1002/ajh.26352 - DOI - PubMed
-
- Arber DA, Orazi A, Hasserjian RP, et al. . International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022;140(11):1200-1228. https://doi.org/10.1182/blood.2022015850 10.1182/blood.2022015850 - DOI - PMC - PubMed
-
- Kim AS, Pozdnyakova O. SOHO State of the Art Updates and Next Questions | Myeloid/Lymphoid Neoplasms with Eosinophilia and Gene Rearrangements: Diagnostic Pearls and Pitfalls. Clin Lymphoma Myeloma Leuk. 2022;22(9):643-651. https://doi.org/10.1016/j.clml.2022.03.008 10.1016/j.clml.2022.03.008 - DOI - PubMed
-
- Zanelli M, Smith M, Zizzo M, et al. . A tricky and rare cause of pulmonary eosinophilia: myeloid/lymphoid neoplasm with eosinophilia and rearrangement of PDGFRA. BMC Pulm Med. 2019;19(1):216. Published 2019 Nov 19. https://doi.org/10.1186/s12890-019-0967-7 10.1186/s12890-019-0967-7 - DOI - PMC - PubMed
-
- Saft L, Kvasnicka HM, Boudova L, Gianelli U, Lazzi S, Rozman M. Myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase fusion genes: A workshop report with focus on novel entities and a literature review including paediatric cases. Histopathology. 2023;83(6):829-849. https://doi.org/10.1111/his.15021 10.1111/his.15021 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

