Terahertz Mie scattering in tissue: diffuse polarimetric imaging and Monte Carlo validation in highly attenuating media models
- PMID: 40474939
- PMCID: PMC12135335
- DOI: 10.1117/1.JBO.30.6.066001
Terahertz Mie scattering in tissue: diffuse polarimetric imaging and Monte Carlo validation in highly attenuating media models
Abstract
Significance: Changes in the structure of tissue occur in many disease processes, such as the boundaries of cancerous tumors and burn injuries. Spectroscopic and polarimetric alterations of terahertz light caused by Mie scattering patterns have the potential to be a diagnostic marker.
Aim: We present an analysis of Monte Carlo simulation of Mie scattering of polarized terahertz light from cancerous tumor budding, compare the simulation with experimental results obtained in phantom models, and present an analysis of a polarization-sensitive terahertz scan of an ex vivo porcine burn injury.
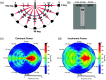
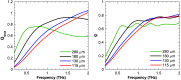
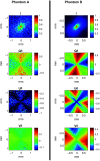
Approach: Using a Monte Carlo simulation, we modeled the changes in diffuse intensity and degree of polarization of broadband off-specular terahertz light due to scattering particles in highly attenuating tissue. We extracted the Mueller matrix of the tissue using this model and analyzed the Lu-Chipman product decomposition matrices. We compared this model with experimental data from four phantoms consisting of polypropylene particles of varying sizes embedded in gelatin. Finally, we induced a full-thickness burn injury in ex vivo porcine skin samples and compared experimental data captured over burned and healthy regions of the tissue.
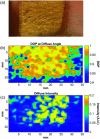
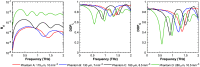
Results: Simulation revealed contrast in the Stokes vectors and Mueller Matrix elements for varying scattering particle sizes. Experimental phantom results showed contrast between different sizes of scattering particles in degree of polarization and diffuse intensity in agreement with Monte Carlo simulation results. Finally, we demonstrated a similar diffused imaging signal contrast between burned and healthy regions of ex vivo porcine skin.
Conclusion: Polarimetric terahertz imaging has the potential to detect structural changes due to biological disease processes.
Keywords: Mie scattering; degree of polarization; diffused scattering; terahertz time-domain spectroscopic imaging; terahertz time-domain spectroscopy and polarimetry; tumor budding.
© 2025 The Authors.
Figures




Similar articles
-
Structured light imaging mesoscopy: detection of embedded morphological changes in superficial tissues.J Biomed Opt. 2025 Jun;30(6):065001. doi: 10.1117/1.JBO.30.6.065001. Epub 2025 Jun 18. J Biomed Opt. 2025. PMID: 40534904 Free PMC article.
-
Polarimetry terahertz imaging of human breast cancer surgical specimens.J Med Imaging (Bellingham). 2024 Nov;11(6):065503. doi: 10.1117/1.JMI.11.6.065503. Epub 2024 Dec 5. J Med Imaging (Bellingham). 2024. PMID: 39649775
-
Mueller matrix analysis of a biologically sourced engineered tissue construct as polarimetric phantom.J Biomed Opt. 2024 Oct;29(10):106002. doi: 10.1117/1.JBO.29.10.106002. Epub 2024 Oct 29. J Biomed Opt. 2024. PMID: 39474362 Free PMC article.
-
Magnetic resonance perfusion for differentiating low-grade from high-grade gliomas at first presentation.Cochrane Database Syst Rev. 2018 Jan 22;1(1):CD011551. doi: 10.1002/14651858.CD011551.pub2. Cochrane Database Syst Rev. 2018. PMID: 29357120 Free PMC article.
-
The value of FDG positron emission tomography/computerised tomography (PET/CT) in pre-operative staging of colorectal cancer: a systematic review and economic evaluation.Health Technol Assess. 2011 Sep;15(35):1-192, iii-iv. doi: 10.3310/hta15350. Health Technol Assess. 2011. PMID: 21958472 Free PMC article.
References
-
- Leitenstorfer A., et al. , “The 2023 terahertz science and technology roadmap,” J. Phys. D: Appl. Phys. 56(22), 223001 (2023).JPAPBE10.1088/1361-6463/acbe4c - DOI
-
- Chen X., et al. , “Terahertz (THz) biophotonics technology: instrumentation, techniques, and biomedical applications,” Chem. Phys. Rev. 3(1), 011311 (2022).10.1063/5.0068979 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

