This is a preprint.
Adaptive Machine Learning Framework enables Unprecedented Yield and Purity of Adeno-Associated Viral Vectors for Gene Therapy
- PMID: 40475650
- PMCID: PMC12139824
- DOI: 10.1101/2025.05.23.655859
Adaptive Machine Learning Framework enables Unprecedented Yield and Purity of Adeno-Associated Viral Vectors for Gene Therapy
Update in
-
Adaptive Machine Learning Framework for Optimizing the Affinity Purification of Adeno-Associated Viral Vectors.Biotechnol Bioeng. 2026 Jan 19. doi: 10.1002/bit.70159. Online ahead of print. Biotechnol Bioeng. 2026. PMID: 41553185
Abstract
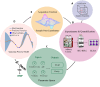
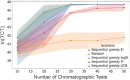
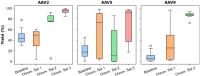
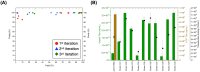
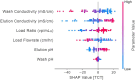
Adeno-associated viral (AAV) vectors for gene therapy are becoming integral to modern medicine, providing therapeutic options for diseases once deemed incurable. Currently, optimizing viral vector purification is a critical bottleneck in the gene therapy industry, impacting product efficacy and safety as well as accessibility and cost to patients. Traditional optimization methods are resource-intensive and often fail to adjust the purification process parameters to maximize the resulting product yield and quality. To address this challenge, we developed a machine learning framework that leverages Bayesian optimization to systematically refine affinity chromatography parameters (sample load, flow rate, and the formulation of chromatographic media) to improve AAV purification. The efficiency of this closed-loop workflow in iteratively optimizing the vector's yield, purity, and transduction efficiency was demonstrated by purifying clinically-relevant serotypes AAV2, AAV5, and AAV9 from HEK293 cell lysates using the affinity adsorbent AAVidity. We show that three cycles of Bayesian optimization elevated yields from a baseline of 70% to 99%, while reducing host-cell impurities by 230-to-400-fold across all serotypes. The optimized parameters consistently produced vectors with high purity and preserved high transduction activity, essential for therapeutic efficacy and safety, demonstrating serotype versatility - a key challenge in AAV manufacturing. By streamlining parameter optimization and enhancing productivity, our adaptive machine learning framework accelerates process development and reduces costs, advancing the accessibility and clinical translation of AAV-based gene therapies.
Keywords: Bayesian optimization; Gene therapy; machine learning; purification; viral vectors.
Conflict of interest statement
Conflict of interest. AT and ENM are co-founders of Predictive, LLC, which develops novel alternative methods and software for toxicity prediction. SM is Chief Technology Officer of LigaTrap Technology, which commercializes the AAVidity affinity resin. All other authors have no conflict of interest to disclose.
Figures


References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
