Knockout of bcas3 gene causes neurodevelopment defects in zebrafish
- PMID: 40481608
- PMCID: PMC12142951
- DOI: 10.1186/s40659-025-00615-4
Knockout of bcas3 gene causes neurodevelopment defects in zebrafish
Abstract
Background: Neurodevelopmental disorders manifest in early childhood and are characterized by cognitive deficits, intellectual disabilities, motor disorders, and social dysfunction. Mutations in BCAS3 gene are associated with syndromic neurodevelopmental disorders in humans, while the detailed pathological mechanism is still unknown.
Methods: CRISPR/Cas9 technology was used to generate a bcas3 knockout zebrafish model. To investigate the effects of bcas3 on development, morphological evaluations were conducted. Locomotor behaviors, including performance in the light-dark test, novel tank test, mirror test, shoaling test, and social test, were assessed through video tracing and quantitative analysis of movement parameters. Transcriptome sequencing analysis was used to identify dysregulated pathways associated with development process. Additionally, Acridine Orange staining was employed to evaluate apoptosis. Western blot and real-time RT-PCR were used to analyze the expression levels of genes.
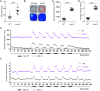
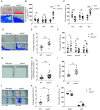
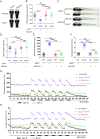
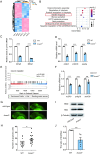
Results: Bcas3 knockout zebrafish exhibited early larval phenotypes resembling clinical features of patients with BCAS3 mutations, including global delayed development at early embryonic development, microcephaly and reduced body length. Behavior analysis revealed abnormal motor dysfunction, such as social impairment, increased anxiety and heightened aggression. Notably, human BCAS3 rescued the developmental defects and motor disorders in bcas3 knockout larvae. Transcriptomic analysis identified substantial downregulation of genes related to embryonic development and startle response, brain development and neuron migration in bcas3 knockout zebrafish, such as rpl10, cyfip2, erbb3b, eya4a, nr2f1b, prkg1b and ackr3b. Additionally, increased apoptosis was observed in bcas3 knockout zebrafish, which was further confirmed by Acridine Orange staining and a decreased Bcl2/Bax ratio in western blot analysis. The increased apoptosis observed in the brain of bcas3 knockout larvae could contribute to the developmental and locomotor deficits.
Conclusion: The bcas3 knockout zebrafish model recapitulates the clinical features observed in patients with BCAS3 mutations. Our results suggest that increased apoptosis may underlie the developmental deficits and motor disorders in these patients. The bcas3 knockout zebrafish model provides a valuable tool to identify dysregulated molecular targets for therapeutic intervention during the early stages of disease progression.
Supplementary Information: The online version contains supplementary material available at 10.1186/s40659-025-00615-4.
Keywords: BCAS3; CRISPR/Cas9; Neurodevelopmental disorder; Zebrafish.
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures


References
-
- Stamberger H, Nikanorova M, Willemsen MH, Accorsi P, Angriman M, Baier H, Benkel-Herrenbrueck I, Benoit V, Budetta M, Caliebe A, Cantalupo G, Capovilla G, Casara G, Courage C, Deprez M, Destrée A, Dilena R, Erasmus CE, Fannemel M, Fjær R, Giordano L, Helbig KL, Heyne HO, Klepper J, Kluger GJ, Lederer D, Lodi M, Maier O, Merkenschlager A, Michelberger N, Minetti C, Muhle H, Phalin J, Ramsey K, Romeo A, Schallner J, Schanze I, Shinawi M, Sleegers K, Sterbova K, Syrbe S, Traverso M, Tzschach A, Uldall P, Van Coster R, Verhelst H, Viri M, Winter S, Wolff M, Zenker M, Zoccante L, De Jonghe P, Helbig I, Striano P, Lemke JR, Møller RS, S., Weckhuysen. STXBP1 encephalopathy: A neurodevelopmental disorder including epilepsy. Neurology 2016;86(10): 954– 62. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

