Exploring the correlation and mechanism of natural killer cell cytotoxic sensitivity against gastric cancer
- PMID: 40486876
- PMCID: PMC12144607
- DOI: 10.32604/or.2025.059426
Exploring the correlation and mechanism of natural killer cell cytotoxic sensitivity against gastric cancer
Abstract
Background: Human natural killer (NK) cells have attracted widespread attention as a potential adoptive cell therapy (ACT). However, the therapeutic effects of NK cell infusion in patients with solid tumors are limited. There is an urgent need to explore a suitable new treatment plan to overcome weaknesses and support the superior therapeutic activity of NK cells.
Methods: In this study, the mechanisms underlying the susceptibility of gastric cancer (GC) cell lines AGS, HGC-27, and NCI-N87 to NK cell-mediated cytotoxicity were explored.
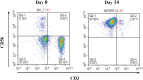
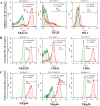
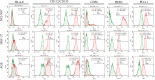
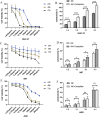
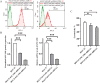
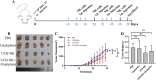
Results: Lactic dehydrogenase (LDH) release assays showed that all three GC cell lines were susceptible to the umbilical cord blood NK (UCB-NK) cells, and HGC-27 cells with high CD56 expression were the most sensitive to UCB-NK, followed by NCI-N87 and AGS. When the expression of CD56 in HGC-27 cells decreased, the lytic activity of NK cells in HGC-27 cells was abating. In addition, combining oxaliplatin with NK cells produced additive anti-tumor effects in vitro, which may have resulted from oxaliplatin-induced NK group 2 member D (NKG2DL) upregulation in GC cells. These results of cytotoxicity activity showed that inhibition of CD56 expression might suppress the sensitivity of GC cells to NK cell-mediated cytotoxicity, and upregulation of the expression of NKG2DL on the surface of GC cells by oxaliplatin could enhance the killing sensitivity of NK cells.
Conclusion: Collectively, our study provides a deeper theoretical foundation and a better therapeutic strategy for NK cell immunotherapy in the treatment of human GC.
Keywords: CD56 (neural cell adhesion molecule NCAM); Gastric cancer; Natural killer group 2 member D (NKG2D) ligand (NKG2DL); Oxaliplatin; Umbilical cord blood natural killer (UCB-NK) cells.
© 2025 The Authors.
Conflict of interest statement
The authors declare no conflict of interest to report regarding the present study.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
