Enhanced dry reforming of methane over nickel catalysts supported on zirconia coated mesoporous silica
- PMID: 40487445
- PMCID: PMC12145839
- DOI: 10.1016/j.isci.2025.112582
Enhanced dry reforming of methane over nickel catalysts supported on zirconia coated mesoporous silica
Abstract
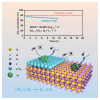
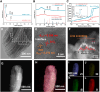
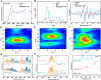
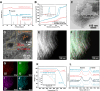
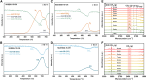
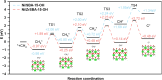
Dry reforming of methane (DRM) offers a sustainable route to convert CH4 and CO2 into syngas, addressing both greenhouse gas emissions and energy demand. However, catalyst deactivation due to sintering and coking limits practical applications. In this work, we developed a mesoporous Ni-based catalyst (Ni/ZrSBA-15-OH) featuring abundant Ni-ZrO2 interfaces and small Ni nanoparticles (5.6 nm) confined within a stable silica framework. This catalyst showed excellent performance, achieving 80% CH4 and 87% CO2 conversions at 750°C, with minimal coke formation (0.4 mg gcat -1 h-1) and high durability (1.3% CH4 conversion loss over 20 h). Advanced characterizations (X-ray absorption spectroscopy [XAS], transmission electron microscopy [TEM], H2-temperature programmed reduction [H2-TPR], and temperature-programmed surface reaction [TPSR]) revealed that the metal-oxide interface enhances the activation of reactants and stabilizes active sites. Density functional theory (DFT) calculations confirmed that the Ni-ZrO2 interface increases the energy barrier for CH∗ dehydrogenation, effectively suppressing carbon deposition. This study provides a rational strategy for designing structurally robust and coke-resistant Ni-based catalysts for efficient DRM.
Keywords: Chemistry; Materials chemistry; Materials science.
© 2025 The Author(s).
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
LinkOut - more resources
Full Text Sources
Miscellaneous

