Estimated cost-effectiveness of early screening strategies for newborn hearing impairment using a Markov model
- PMID: 40487536
- PMCID: PMC12141332
- DOI: 10.3389/fpubh.2025.1498860
Estimated cost-effectiveness of early screening strategies for newborn hearing impairment using a Markov model
Abstract
Background: Decision-making on how to conduct the concurrent hearing and genetic screening of newborns lacks a health economics basis. To estimate the cost-effectiveness of different newborn hearing screening strategy is necessary.
Methods: A decision tree for a simulated cohort population of 9.56 million newborns was developed: (1) Only universal newborn hearing screening (UNHS); (2) Targeted deafness genetic screening; (3) Concurrent hearing and genetic screening. Markov model was used to evaluate the lifetime horizon (78 years). Cost values were estimated based on medical expenses at Beijing Tongren Hospital and previous studies based on a health system perspective. Health state utility values were represented by Quality-adjusted Life Years (QALYs), costs and utilities were discounted at a rate of 3%. The Incremental Cost-effectiveness Ratio (ICER) was analyzed, along with one-way sensitivity analysis and probability sensitivity analysis.
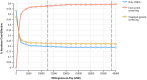
Results: Compared with only UNHS strategy, the ICER of targeted screening strategy is $ 181.9/QALY, the ICER of concurrent screening is $ 1,563.45/QALY at the discounted rate of 3%. UNHS confirms 21,098 cases of hearing loss, of which 18,666 patients receive early hearing intervention. Concurrent screening confirms 34,244 cases of hearing loss, 26,000 of which receive early hearing intervention. Additionally, 27,036 cases of ototoxicity deafness are avoided. Reducing the cost of genetic screening could enhance the cost-effectiveness of both concurrent screening strategy and targeted genetic screening strategies. At a willingness to pay threshold of 1 time the per capita GDP, at $12,741, the probability of the concurrent screening strategy being cost-effective was 57.6%; at a willingness to pay is three times the per capita GDP, at $38,223, the probability of the concurrent screening strategy being cost-effective was 59.10%.
Conclusion: Both the targeted screening and concurrent screening strategy exhibit good cost-effectiveness compared with only UNHS strategy. The targeted screening strategy is preferable when the willingness to pay is between $181.90/QALY and $1,563.45/QALY, whereas the concurrent screening strategy becomes the preferred choice when the willingness to pay exceeds $1,563.45/QALY.
Keywords: Markov model; cost-effectiveness; deafness; genetic screening; newborn hearing screening.
Copyright © 2025 Wen, Wang, Shu, Ruan, Xie, Cheng, Qi, En, Qin, Huang and Han.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

