Effect of ranibizumab on diabetic retinopathy via the vascular endothelial growth factor/STAT3/glial fibrillary acidic protein pathway
- PMID: 40487605
- PMCID: PMC12142178
- DOI: 10.4239/wjd.v16.i5.99473
Effect of ranibizumab on diabetic retinopathy via the vascular endothelial growth factor/STAT3/glial fibrillary acidic protein pathway
Abstract
Background: Diabetic retinopathy (DR) is the leading cause of vision loss in patients with diabetes. The vascular endothelial growth factor (VEGF) pathway plays a critical role in the pathogenesis of DR, and ranibizumab, an anti-VEGF agent, has shown promise in its treatment. Signal transducer and activator of transcription 3 (STAT3) is involved in inflammatory processes and cellular signaling, while glial fibrillary acidic protein (GFAP) is a marker of glial cell activation, both contributing to retinal damage in DR. However, the mechanisms by which ranibizumab affect early-stage DR through the VEGF/STAT3/GFAP pathway are not fully understood.
Aim: To investigate the role of ranibizumab in early DR via the VEGF/STAT3/GFAP pathway.
Methods: Adult retinal pigment epithelial 19 (ARPE-19) cells and human retinal microvascular endothelial cells (HRMECs) were cultured under high-glucose conditions to simulate a diabetic environment. The effects of ranibizumab on cytokine mRNA and protein expression were analyzed by quantitative polymerase chain reaction and Western blot analysis. A diabetic rat model was induced with streptozotocin (60 mg/kg). Retinal changes, including retinal ganglion cell (RGC) apoptosis, vascular alterations, and cytokine expression, were evaluated using fundus fluorescein angiography, hematoxylin and eosin and periodic acid Schiff staining, immunofluorescence, confocal imaging, and Western blot analysis.
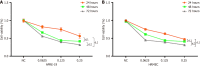
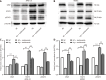
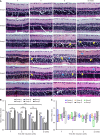
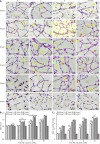
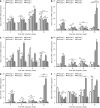
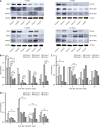
Results: High-glucose conditions significantly increased the mRNA and protein levels of VEGF, STAT3, GFAP, and other cytokines in ARPE-19 and HRMECs. However, these levels were partially suppressed by ranibizumab. RGC apoptosis, vascular leakage, and elevated cytokine expression were observed during early-stage DR in diabetic rats. Ranibizumab treatment in diabetic rats reduced cytokine expression, restored RGCs, and repaired vascular networks.
Conclusion: Intravitreal ranibizumab modulates the VEGF/STAT3/GFAP pathway, suppresses cytokine expression, and promotes retinal repair, effectively delaying or preventing early DR progression.
Keywords: Diabetic retinopathy; Early stage; Glial fibrillary acidic protein; Ranibizumab; Signal transducer and activator of transcription 3; Vascular endothelial growth factor.
©The Author(s) 2025. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors have no conflicts of interest to declare.
Figures




Similar articles
-
Effect of intravitreal injection of ranibizumab on retinal ganglion cells and microvessels in the early stage of diabetic retinopathy in rats with streptozotocin-induced diabetes.Exp Ther Med. 2017 Jun;13(6):3360-3368. doi: 10.3892/etm.2017.4431. Epub 2017 May 5. Exp Ther Med. 2017. PMID: 28587414 Free PMC article.
-
Genipin relieves diabetic retinopathy by down-regulation of advanced glycation end products via the mitochondrial metabolism related signaling pathway.World J Diabetes. 2023 Sep 15;14(9):1349-1368. doi: 10.4239/wjd.v14.i9.1349. World J Diabetes. 2023. PMID: 37771331 Free PMC article.
-
Role of the JAK/STAT pathway in a streptozotocin-induced diabetic retinopathy mouse model.Graefes Arch Clin Exp Ophthalmol. 2022 Nov;260(11):3553-3563. doi: 10.1007/s00417-022-05694-7. Epub 2022 May 23. Graefes Arch Clin Exp Ophthalmol. 2022. PMID: 35599279
-
[Cell biology of intraocular vascular diseases].Nippon Ganka Gakkai Zasshi. 1999 Dec;103(12):923-47. Nippon Ganka Gakkai Zasshi. 1999. PMID: 10643294 Review. Japanese.
-
Ranibizumab for the treatment of diabetic retinopathy.Expert Opin Biol Ther. 2021 Aug;21(8):991-997. doi: 10.1080/14712598.2021.1928629. Epub 2021 May 19. Expert Opin Biol Ther. 2021. PMID: 33966556 Review.
References
-
- Kwan CC, Fawzi AA. Imaging and Biomarkers in Diabetic Macular Edema and Diabetic Retinopathy. Curr Diab Rep. 2019;19:95. - PubMed
-
- Antar SA, Ashour NA, Sharaky M, Khattab M, Ashour NA, Zaid RT, Roh EJ, Elkamhawy A, Al-Karmalawy AA. Diabetes mellitus: Classification, mediators, and complications; A gate to identify potential targets for the development of new effective treatments. Biomed Pharmacother. 2023;168:115734. - PubMed
-
- Semeraro F, Morescalchi F, Cancarini A, Russo A, Rezzola S, Costagliola C. Diabetic retinopathy, a vascular and inflammatory disease: Therapeutic implications. Diabetes Metab. 2019;45:517–527. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

