Baoshentongluo Formula relieves podocyte injury in diabetic kidney disease through regulating mitophagy via PINK1/Parkin signaling pathway
- PMID: 40487763
- PMCID: PMC12141005
- DOI: 10.3389/fendo.2025.1606326
Baoshentongluo Formula relieves podocyte injury in diabetic kidney disease through regulating mitophagy via PINK1/Parkin signaling pathway
Abstract
Introduction: Diabetic kidney disease (DKD) progression is strongly associated with podocyte mitochondrial dysfunction. The clinically effective Chinese herbal Baoshentongluo formula (BSTL) has demonstrated significant proteinuria reduction in DKD patients. HPLC-ESI-MS analysis identified characteristic bioactive components in BSTL including astragalosides, rehmanniosides, and tanshinones. However, the molecular mechanisms through which BSTL maintains podocyte homeostasis remain incompletely understood.
Methods: Mouse podocyte clone-5 (MPC-5) cells and db/db mice were used. Db/db mice were randomized into db/db and db/db + BSTL (16.5 g/kg/d, intragastric administration for 12 weeks). A group of m/m mice served as the control. Renal function, urinary albumin-to-creatinine ratio (UACR), histopathological analysis, apoptotic, and mitophagy-related protein levels were evaluated. MPC-5 cells were exposed to high glucose (HG, 30 mM) and BSTL drug-containing serum (8%) for 24 h grouping as control, HG, HG + BSTL, and HG + siPINK1. Podocyte apoptosis, mitophagy levels, and expression of PTEN-induced putative kinase 1 (PINK1) and E3 ubiquitin ligase (Parkin) were assessed.
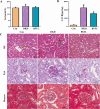
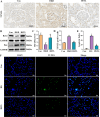
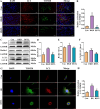
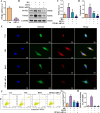
Results: In db/db diabetic mice, oral administration of BSTL significantly lowered urinary albumin-to-creatinine ratio (P<0.05), improved glomerular filtration rate, and ameliorated renal histopathological changes, decreased LC3-II/LC3-I ratio, and downregulated expression of mitophagy-related proteins PINK1, Parkin, ATG5 and Beclin-1. Treatment with 8% BSTL-containing serum significantly attenuated HG-induced podocyte apoptosis (P<0.01) and suppressed excessive mitophagy, as evidenced by reduced TOM20/LC3 co-localization (P<0.01). Notably, BSTL treatment markedly reduced protein levels of both PINK1 and Parkin (P<0.01), key regulators of mitophagy initiation. Genetic silencing of PINK1 in podocytes phenocopied BSTL's protective effects, confirming the pathway specificity.
Discussion: Our integrated in vitro and in vivo findings establish that BSTL protects against DKD progression by selectively inhibiting PINK1/Parkin-dependent mitophagy in podocytes to inhibit podocyte injury, which provides both mechanistic insights and therapeutic potential for clinical DKD management.
Keywords: apoptosis; diabetic kidney disease; mitochondria; podocyte; traditional Chinese medicine.
Copyright © 2025 Pang, Tian, Liu, Guo, Zhao, Wang, Wang and Zhao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

