Ultrasound-Assisted Synthesis of 2‑Benzylidene-1-Indanone Derivatives and Evaluation as a Corrosion Inhibitor for Mild Steel in 1 M HCl Solution
- PMID: 40488013
- PMCID: PMC12138709
- DOI: 10.1021/acsomega.4c09705
Ultrasound-Assisted Synthesis of 2‑Benzylidene-1-Indanone Derivatives and Evaluation as a Corrosion Inhibitor for Mild Steel in 1 M HCl Solution
Abstract
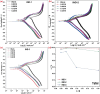
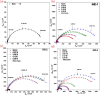
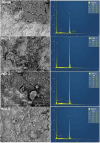
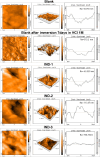
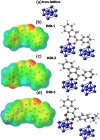
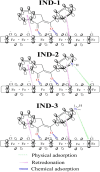
This study explores the ultrasound-assisted synthesis of three 2-benzylidene-1-indanone derivatives and assesses their effectiveness as corrosion inhibitors for mild steel AISI 1018 in 1 M HCl through electrochemical techniques and density functional theory (DFT) quantum chemical calculations. The results indicate that the inhibitors studied; specifically IND-1, (Z)-2-(hydroxy-(phenyl)-methylene)-2,3-dihydro-1H-inden-1-one; IND-2, (Z)-2-(hydroxy-(pyridine-3-yl)-methylene)-2,3-dihydro-1H-inden-1-one and IND-3, (Z)-2-((4-(dimethylamino)-phenyl)-(hydroxy)-methylene)-2,3-dihydro-1H-inden-1-one, modified the cathodic reaction mechanism. These inhibitors achieve efficiencies greater than 98%, and their inhibitory effect does not increase with higher concentrations. A comparison of computational predictions and experimental analyses indicates that the pyridine ring and the diethylamino group reduce the inhibitory capacity of carbon steel in acidic environments.
© 2025 The Authors. Published by American Chemical Society.
Figures
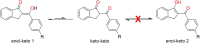


References
-
- Kadhim A., Al-Amiery A., Alazawi R., Al-Ghezi M., Abass R.. Corrosion inhibitors. A review. Int. J. Corrosion Scale Inhibition. 2021;10(1):54–67. doi: 10.17675/2305-6894-2021-10-1-3. - DOI
-
- Chigondo M., Chigondo F.. Recent Natural Corrosion Inhibitors for Mild Steel: An Overview. J. Chem. 2016;2016(1):6208937. doi: 10.1155/2016/6208937. - DOI
-
- Solomon M. M., Umoren S. A.. Enhanced corrosion inhibition effect of polypropylene glycol in the presence of iodide ions at mild steel/sulphuric acid interface. J. Environ. Chem. Eng. 2015;3(3):1812–1826. doi: 10.1016/j.jece.2015.05.018. - DOI
-
- Ansari K., Quraishi M., Singh A.. Schiff’s base of pyridyl substituted triazoles as new and effective corrosion inhibitors for mild steel in hydrochloric acid solution. Corros. Sci. 2014;79:5–15. doi: 10.1016/j.corsci.2013.10.009. - DOI
-
- Saranya J., Sowmiya M., Sounthari P., Parameswari K., Chitra S., Senthilkumar K.. N-heterocycles as corrosion inhibitors for mild steel in acid medium. J. Mol. Liq. 2016;216:42–52. doi: 10.1016/j.molliq.2015.12.096. - DOI
LinkOut - more resources
Full Text Sources
