MYL1-Related Congenital Myopathy: Clinical, Genetic and Pathological Insights
- PMID: 40488356
- PMCID: PMC12147433
- DOI: 10.1111/nan.70025
MYL1-Related Congenital Myopathy: Clinical, Genetic and Pathological Insights
Abstract
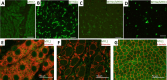
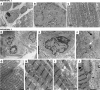
Congenital myopathies and congenital muscular dystrophies encompass heterogeneous clinical and genetic groups of disorders characterised by muscle weakness with antenatal or early postnatal onset. These conditions are categorised according to distinctive myopathological features and causative genes. Despite advances in diagnosis through massive parallel sequencing and progress in understanding the underlying pathogenesis, many aspects of these disorders remain poorly understood. MYL1-related congenital myopathy is an ultra-rare and severe condition, associated with a deficiency of essential/alkali light myosin and impaired development of fast-twitch type II muscle fibres. This study aims to advance the understanding of the phenotype and pathogenesis of MYL1-congenital myopathy. We analysed the clinical characteristics of two individuals harbouring three novel variants in the MYL1 gene. We conducted detailed genomic analysis and extensive studies on their muscles using histological, immunohistochemical, immunofluorescence, Western Blot and electron microscopy. Both individuals showed a very severe congenital myopathy, characterised by congenital hypotonia and weakness, requiring ventilatory and nutritional assistance. Muscle biopsy revealed dystrophic-like or myopathic changes, with notable smallness of fast-twitch type II fibres, often arranged around larger type I fibres, drawing a floret pattern. These fibres expressed developmental myosin and exhibited features of aberrant myofibrillogenesis. Type I myofibres exhibited correct sarcomere alignment, but like the small fast-twitch fibres, both showed distorted cell organelles, vacuolar aggregates and membranous debris, indicating autophagic impairment. Our findings confirm that bi-allelic MYL1 variants are associated with a severe congenital myopathy, characterised by a distinctive clinical and histopathological phenotype involving impaired type II fibre development. Additionally, our study reveals a role for MYL1 in the organisation and trophism of all muscle fibre types. SUMMARY: MYL1 biallelic variants cause severe congenital myopathy with early hypotonia and type II fibre hypotrophy. Muscle biopsy shows a distinct pattern, including floret-like fibre arrangement. Findings suggest a broader role for MYL1 in fibre organisation and autophagy across muscle fibre types.
Keywords: congenital muscular dystrophy; congenital myopathy; essential/alkali myosin light chain; fast‐twitch type II muscle fibres; muscle cell autophagy; myosin heavy chain.
© 2025 The Author(s). Neuropathology and Applied Neurobiology published by John Wiley & Sons Ltd on behalf of British Neuropathological Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

