Mutations associated with resistance to rifampicin and isoniazid identified in strains of the Mycobacterium tuberculosis complex by GenoType MTBDRplus in Panama, 2015-2021
- PMID: 40488462
- PMCID: PMC12211072
- DOI: 10.1128/spectrum.02400-24
Mutations associated with resistance to rifampicin and isoniazid identified in strains of the Mycobacterium tuberculosis complex by GenoType MTBDRplus in Panama, 2015-2021
Abstract
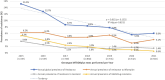
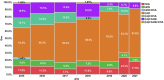
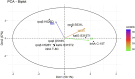
Tuberculosis is one of the diseases causing high rates of morbidity and mortality in several countries. However, efforts in the use of diagnostic methods for tuberculosis and the detection of drug resistance are essential to reduce cases. Since 2015, rapid molecular diagnostic tests have been implemented in Panama, enabling the detection of drug resistance, mainly rifampicin and isoniazid, in patients with suspected tuberculosis. This study aimed to identify mutations in Mycobacterium tuberculosis complex strains with resistance to rifampicin and isoniazid using GenoType MTBDRplus. It is a retrospective study reviewing the results of the GenoType MTBDRplus version 2.0 test from 2015 to 2021. Strains not identified as Mycobacterium tuberculosis complex and those that did not show a mutation pattern and were categorized as sensitive were excluded. Data analysis was carried out using the Chi-square tests, Pearson's Correlation, and principal components analysis. A total of 4,301 strains were analyzed, of which 8.8% were detected with mutation or resistance probes in one or more of the genes analyzed: 56.0% in the rpoβ gene, 11.9% in the inhA gene, and 8.2% in the katG gene. In addition, other mutations such as rpoβ/inhA and rpoβ/katG were detected in 9.5% and 13.5% of cases, respectively. Thirty-eight resistance patterns were identified, with H526D and S531L being the most frequent mutations in the rpoB gene, and S315T1 and C-15T are the most common in katG and inhA, respectively. The resistance patterns detected by the GenoType MTBDRplus assay highlight the genetic variability of drug resistance in the country and emphasize the need to implement epidemiological surveillance methodologies. Integrating patient clinical data with genetic variation information is essential for improving disease control and understanding transmission dynamics and drug resistance acquisition. These findings also provide important insights for guiding tuberculosis treatment strategies in Panama, supporting the use of molecular tools for the early detection of drug resistance, enhancing our understanding of the epidemiology, and informing clinical decision-making.
Importance: This study focuses on understanding how Mycobacterium tuberculosis strains in Panama develop resistance. With tuberculosis (TB) cases becoming harder to treat due to drug resistance, especially after the disruptions caused by the COVID-19 pandemic, rapid and accurate diagnosis is crucial. By using advanced molecular tests to identify specific genetic mutations in drug-resistant TB strains, this research helps improve treatment decisions, leading to better outcomes for patients. Understanding these mutations also aids in controlling the spread of TB. Given the rising global concern over drug-resistant TB, the findings of this study are important not only for Panama but also for other regions facing similar challenges.
Keywords: Mycobacterium tuberculosis; Panama; drug resistance; genotype; mutation; tuberculosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- World Health Organization . 2022. Global tuberculosis report 2022. Geneva.
-
- Organización Panamericana de la Salud . 2020. Comunicación rápida: análisis moleculares como pruebas diagnósticas iniciales de la tuberculosis y la resistencia a la rifampicina
-
- Organización Panamericana de la Salud . 2021. Actualización sobre el uso de pruebas de amplificación de ácidos nucleicos para detectar la tuberculosis y la tuberculosis farmacorresistente: comunicación rápida
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

