Neuroserpin alleviates cerebral ischemia-reperfusion injury by suppressing ischemia-induced endoplasmic reticulum stress
- PMID: 40489346
- PMCID: PMC12094565
- DOI: 10.4103/NRR.NRR-D-24-00044
Neuroserpin alleviates cerebral ischemia-reperfusion injury by suppressing ischemia-induced endoplasmic reticulum stress
Abstract
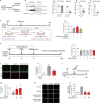
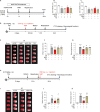
JOURNAL/nrgr/04.03/01300535-202601000-00037/figure1/v/2025-06-09T151831Z/r/image-tiff Neuroserpin, a secreted protein that belongs to the serpin superfamily of serine protease inhibitors, is highly expressed in the central nervous system and plays multiple roles in brain development and pathology. As a natural inhibitor of recombinant tissue plasminogen activator, neuroserpin inhibits the increased activity of tissue plasminogen activator in ischemic conditions and extends the therapeutic windows of tissue plasminogen activator for brain ischemia. However, the neuroprotective mechanism of neuroserpin against ischemic stroke remains unclear. In this study, we used a mouse model of middle cerebral artery occlusion and oxygen-glucose deprivation/reperfusion-injured cortical neurons as in vivo and in vitro ischemia-reperfusion models, respectively. The models were used to investigate the neuroprotective effects of neuroserpin. Our findings revealed that endoplasmic reticulum stress was promptly triggered following ischemia, initially manifesting as the acute activation of endoplasmic reticulum stress transmembrane sensors and the suppression of protein synthesis, which was followed by a later apoptotic response. Notably, ischemic stroke markedly downregulated the expression of neuroserpin in cortical neurons. Exogenous neuroserpin reversed the activation of multiple endoplasmic reticulum stress signaling molecules, the reduction in protein synthesis, and the upregulation of apoptotic transcription factors. This led to a reduction in neuronal death induced by oxygen/glucose deprivation and reperfusion, as well as decreased cerebral infarction and neurological dysfunction in mice with middle cerebral artery occlusion. However, the neuroprotective effects of neuroserpin were markedly inhibited by endoplasmic reticulum stress activators thapsigargin and tunicamycin. Our findings demonstrate that neuroserpin exerts neuroprotective effects on ischemic stroke by suppressing endoplasmic reticulum stress.
Keywords: endoplasmic reticulum stress; ischemia-reperfusion injury; neuron; neuronal apoptosis; neuroprotection; neuroserpin; protein synthesis; secretory protein; stroke; transcriptomic analysis.
Copyright © 2024 Neural Regeneration Research.
Conflict of interest statement
Figures




Similar articles
-
Sappanone A Protects Against Inflammation, Oxidative Stress and Apoptosis in Cerebral Ischemia-Reperfusion Injury by Alleviating Endoplasmic Reticulum Stress.Inflammation. 2021 Jun;44(3):934-945. doi: 10.1007/s10753-020-01388-6. Epub 2021 Jan 7. Inflammation. 2021. PMID: 33411101
-
Ginsenoside Rb1 reduced ischemic stroke-induced apoptosis through endoplasmic reticulum stress-associated IRE1/TRAF2/JNK pathway.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jan;398(1):747-764. doi: 10.1007/s00210-024-03292-4. Epub 2024 Jul 25. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39052059
-
γ-Glutamylcysteine Alleviates Ischemic Stroke-Induced Neuronal Apoptosis by Inhibiting ROS-Mediated Endoplasmic Reticulum Stress.Oxid Med Cell Longev. 2021 Nov 16;2021:2961079. doi: 10.1155/2021/2961079. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 34824669 Free PMC article.
-
Neuroserpin: a selective inhibitor of tissue-type plasminogen activator in the central nervous system.Thromb Haemost. 2004 Mar;91(3):457-64. doi: 10.1160/TH03-12-0766. Thromb Haemost. 2004. PMID: 14983220 Review.
-
Tissue plasminogen activator-independent roles of neuroserpin in the central nervous system.Neural Regen Res. 2012 Jan 15;7(2):146-51. doi: 10.3969/j.issn.1673-5374.2012.02.012. Neural Regen Res. 2012. PMID: 25767491 Free PMC article. Review.
Cited by
-
Impaired Flow: Glymphatic Dysfunction in Ischemic Stroke and the Influence of Sex.J Mol Neurosci. 2025 Jul 4;75(3):85. doi: 10.1007/s12031-025-02377-w. J Mol Neurosci. 2025. PMID: 40615764 Review.
-
Exploring NMDAR pathways in ischemic stroke: implications for neurotoxic and neuroprotective mechanisms and therapeutic strategies.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jun 10. doi: 10.1007/s00210-025-04357-8. Online ahead of print. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40490524 Review.
References
-
- Adorjan I, Tyler T, Bhaduri A, Demharter S, Finszter CK, Bako M, Sebok OM, Nowakowski TJ, Khodosevich K, Møllgård K, Kriegstein AR, Shi L, Hoerder-Suabedissen A, Ansorge O, Molnár Z. Neuroserpin expression during human brain development and in adult brain revealed by immunohistochemistry and single cell RNA sequencing. J Anat. 2019;235:543–554. - PMC - PubMed
-
- Badiola N, Penas C, Miñano-Molina A, Barneda-Zahonero B, Fadó R, Sánchez-Opazo G, Comella JX, Sabriá J, Zhu C, Blomgren K, Casas C, Rodríguez-Alvarez J. Induction of ER stress in response to oxygen-glucose deprivation of cortical cultures involves the activation of the PERK and IRE-1 pathways and of caspase-12. Cell Death Dis. 2011;2:e149. - PMC - PubMed
-
- Barker-Carlson K, Lawrence DA, Schwartz BS. Acyl-enzyme complexes between tissue-type plasminogen activator and neuroserpin are short-lived in vitro. J Biol Chem. 2002;277:46852–46857. - PubMed
-
- Boyce M, Bryant KF, Jousse C, Long K, Harding HP, Scheuner D, Kaufman RJ, Ma D, Coen DM, Ron D, Yuan J. A selective inhibitor of eIF2alpha dephosphorylation protects cells from ER stress. Science. 2005;307:935–939. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

