Anti-Toxoplasma gondii efficacy of beta, beta-dimethylacrylshikonin and isobutyrylshikonin in vitro and in vivo
- PMID: 40490809
- PMCID: PMC12150545
- DOI: 10.1186/s13071-025-06865-1
Anti-Toxoplasma gondii efficacy of beta, beta-dimethylacrylshikonin and isobutyrylshikonin in vitro and in vivo
Abstract
Background: Toxoplasma gondii is a widespread parasite that can infect almost all vertebrate species including humans, causing variable clinical symptoms from asymptomatic infection to serious diseases. Though extensive research has been done in recent decades, the prevention and control of T. gondii continue to present substantial challenges. Herbal medicines have long been a rich source of chemical entities and may provide new avenues for drug discovery against T. gondii. Thus, this study was performed to investigate the anti-T. gondii effect of two monomers, beta, beta-dimethylacrylshikonin (DMAS) and isobutyrylshikonin (IBS), extracted from the roots of a widely distributed and used medical plant.
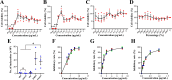
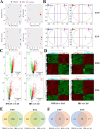
Methods: The cytotoxicity of DMAS and IBS on Vero cells was evaluated using the MTT assay, and the toxicity in mice was assessed on the basis of the changes of body weight combined with the histopathologic examinations on spleen, liver, and kidney. The effects of DMAS and IBS on mice against T. gondii acute infection were evaluated by combining survival curves with splenic histopathologic examination. Ultrastructural change in T. gondii tachyzoites post co-incubation in vitro was observed by electron microscopy. ACT1-quantitative polymerase chain reaction (qPCR) was conducted to quantify T. gondii tachyzoites, including proliferation and the inhibitory efficacy of DMAS and IBS. Invasion and attachment, intracellular proliferation, and parasitophorous vacuole viability evaluations were conducted to assess the effects on the asexual life cycle of T. gondii. In addition, untargeted metabolomics analysis was performed to clarify the underlying mechanisms by which DMAS and IBS act against this parasite.
Results: Both DMAS and IBS, with higher half-maximal cytotoxic concentration (CC50) values, exhibited concentration-dependent cytotoxicity in Vero cells and significantly inhibited the intracellular proliferation of T. gondii in vitro, showing lower half-maximal inhibitory concentration (IC50) values and higher selectivity index (SI) values. DMAS showed a statistically more potent effect than IBS, but both were not significantly more potent than that of pyrimethamine (PM). The tachyzoites exhibited severe ultrastructural damage following treatment with DMAS or IBS. Metabolomics analysis indicated that this abnormal biological lesion was caused by the disruptions in purine and pyrimidine metabolism pathways in T. gondii, with mechanisms likely differing from that of PM. In vivo, a dose of 1.5 mg/kg of DMAS showed no significant toxicity in Kunming (KM) mice, with no significant pathological damage or weight loss. At this dosage, both DMAS and IBS significantly alleviated the splenic hyperemia and statistically prolonged the survival times of T. gondii-infected mice.
Conclusions: This study demonstrated that DMAS and IBS have an inhibitory effect on T. gondii infection in vitro and in vivo, probably associated with the disruption of nucleotide metabolism in the parasite. These results highlight that the two monomers, in particular DMAS, hold promise as a potential therapeutic medicine for toxoplasmosis.
Keywords: Toxoplasma gondii; Anti-infection; Beta, beta-dimethylacrylshikonin; Isobutyrylshikonin; Metabolomics.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All the animals were strictly handled in accordance with the Animal Ethics Procedures and Guidelines of the People’s Republic of China, following established good animal practices. The animal experiments performed in this study were approved by the Experimental Animal Ethics Committee of Guilin Medical University (approval no. GLMC202307169). Consent for publication: Not applicable. Competing interests: Xing-Quan Zhu is a Subject Editor for Parasites & Vectors and was not involved in the peer review of this paper. The authors declare no other competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
- RFSXIHLT202101/the Research Fund of Shanxi Province for Introduced High-level Leading Talents
- 2021XG001/the Special Research Fund of Shanxi Agricultural University for High-level Talents
- 2025GXNSFHA069261/the Natural Science Fund of Guangxi Zhuang Autonomous Region
- 2025KY0536/the Basic Ability Improvement Project for Guangxi Young- and Middle-Aged Teacher Research in University
- GXJKKJ24C009/the Research Project for Disease Control and Prevention in Guangxi
LinkOut - more resources
Full Text Sources
Medical

