Ecophysiological behavior of major Fusarium species in response to combinations of temperature and water activity constraints
- PMID: 40492737
- PMCID: PMC12285238
- DOI: 10.1128/aem.01832-24
Ecophysiological behavior of major Fusarium species in response to combinations of temperature and water activity constraints
Abstract
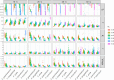
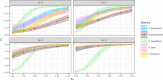
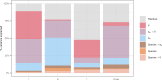
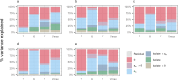
Fusarium head blight (FHB) is a devastating fungal disease affecting cereals, caused by Fusarium species that can produce harmful mycotoxins. Fusarium species coexist within the same ecological niche during infection, with their population dynamics and associated mycotoxin patterns strongly influenced by the environment. This study provides a comprehensive investigation of the ecophysiological responses of the major Fusarium species causing FHB under varying abiotic factors. We assessed growth and mycotoxin production of different isolates of Fusarium avenaceum, Fusarium graminearum, Fusarium langsethiae, Fusarium poae, and Fusarium tricinctum under 24 combinations of temperature (θ = 15, 20, 25, 30°C) and water activity levels (aw = 0.99, 0.98, 0.97, 0.96, 0.95, 0.94). Our findings indicated that θ, aw, and their interaction have a main significant impact on species behavior. Thanks to innovative statistical approaches using fungal growth data from optical density measurements and mycotoxin quantification, we demonstrated significant inter- and intra-specific differences in environmental responses. Growth and mycotoxin production of F. graminearum and F. avenaceum appeared favored under high temperature (≥25°C) and high water activity (≥0.97), whereas lower aw levels (≥0.95) were also conducive for F. poae and F. tricinctum. A specific and unique behavior of F. langsethiae to lowest temperatures (≤20°C) was highlighted. Understanding the ecophysiological requirements of Fusarium species is crucial in the context of climate change, which is expected to worsen disease outbreaks. This study provides valuable knowledge for improving the reliability and robustness of FHB prediction models and anticipating the associated mycotoxin risk.IMPORTANCEFusarium species pose a significant threat to major cereal crops, particularly wheat, by reducing yields and producing mycotoxins that are harmful to animals and humans. The prevalence of each Fusarium species is strongly influenced by environmental conditions, and climate changes have already been reported as responsible for shifts in pathogen populations, leading to changes in mycotoxin patterns. This study revealed distinct ecophysiological behaviors, including growth and mycotoxin production, of the five major Fusarium species infecting small grain cereals when exposed to varying temperature and water activity conditions. Our findings provide a valuable foundation for a deeper understanding of mycotoxin risk and for developing more effective mitigation strategies in the near future.
Keywords: adaptive response; climate change; combined effects; ecological niche; fungal pathogens; inter/intraspecific diversity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Citric acid impairs type B trichothecene biosynthesis of Fusarium graminearum but enhances its growth and pigment biosynthesis: transcriptomic and proteomic analyses.Appl Environ Microbiol. 2025 Jun 18;91(6):e0153124. doi: 10.1128/aem.01531-24. Epub 2025 May 14. Appl Environ Microbiol. 2025. PMID: 40366181 Free PMC article.
-
Comparative analysis of distinct phenotyping methods for assessing wheat resistance and pathogen virulence among Fusarium species causing head blight disease.Plant Methods. 2025 Jun 16;21(1):85. doi: 10.1186/s13007-025-01402-8. Plant Methods. 2025. PMID: 40524223 Free PMC article.
-
Nutritional conditions affecting of selenium nanoparticles synthesized by Fusarium oxysporum (CCASU-2023-F9), and their biological activities against mycotoxin-producing fungi isolated from animal feed.Braz J Microbiol. 2024 Dec;55(4):3465-3476. doi: 10.1007/s42770-024-01494-9. Epub 2024 Sep 6. Braz J Microbiol. 2024. PMID: 39240496
-
Recent insights into potato dry rot an emerging Disease: Focusing on pathogen diversity, host-pathogen interactions, and management strategies.Microb Pathog. 2025 Oct;207:107866. doi: 10.1016/j.micpath.2025.107866. Epub 2025 Jul 9. Microb Pathog. 2025. PMID: 40645351 Review.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
-
- Xu X-M, Parry DW, Nicholson P, Thomsett MA, Simpson D, Edwards SG, Cooke BM, Doohan FM, Brennan JM, Moretti A, Tocco G, Mule G, Hornok L, Giczey G, Tatnell J. 2005. Predominance and association of pathogenic fungi causing Fusarium ear blightin wheat in four European countries. Eur J Plant Pathol 112:143–154. doi: 10.1007/s10658-005-2446-7 - DOI
-
- Beccari G, Colasante V, Tini F, Senatore MT, Prodi A, Sulyok M, Covarelli L. 2018. Causal agents of Fusarium head blight of durum wheat (Triticum durum Desf.) in central Italy and their in vitro biosynthesis of secondary metabolites. Food Microbiol 70:17–27. doi: 10.1016/j.fm.2017.08.016 - DOI - PubMed
-
- Vogelgsang S, Beyer M, Pasquali M, Jenny E, Musa T, Bucheli TD, Wettstein FE, Forrer H-R. 2019. An eight-year survey of wheat shows distinctive effects of cropping factors on different Fusarium species and associated mycotoxins. Eur J Agron 105:62–77. doi: 10.1016/j.eja.2019.01.002 - DOI
-
- Valverde-Bogantes E, Bianchini A, Herr JR, Rose DJ, Wegulo SN, Hallen-Adams HE. 2020. Recent population changes of Fusarium head blight pathogens: drivers and implications. Canadian Journal of Plant Pathology 42:315–329. doi: 10.1080/07060661.2019.1680442 - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

