Drug-associated abdominal aortitis and retroperitoneal fibrosis after treatment with nivolumab
- PMID: 40496660
- PMCID: PMC12151223
- DOI: 10.1016/j.jvscit.2025.101824
Drug-associated abdominal aortitis and retroperitoneal fibrosis after treatment with nivolumab
Abstract
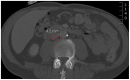
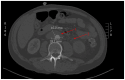
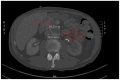
A 60-year-old man with metastatic supraglottic laryngeal cancer treated with nivolumab, an immune checkpoint inhibitor (ICI), developed abdominal aortitis and retroperitoneal fibrosis. The nivolumab was discontinued and no steroids were initiated. A literature search found 19 other cases of ICI-associated aortitis. The evidence is limited and conflicting on the optimal management of these rare but serious adverse events. With increasing application in the treatment of a multitude of cancer types, physicians must remain aware of aortitis as an adverse event and consider the potential risks vs benefits before initiating ICI treatment for their patient.
Keywords: Aortitis; Immune checkpoint inhibitors; Immunotherapy; Retroperitoneal fibrosis.
© 2025 The Authors.
Conflict of interest statement
None.
Figures
Similar articles
-
Aortic rupture following acute aortitis in a patient with head and neck carcinoma treated with nivolumab: a rare but severe immune-related adverse event.Eur Arch Otorhinolaryngol. 2024 Apr;281(4):2037-2040. doi: 10.1007/s00405-024-08495-2. Epub 2024 Feb 3. Eur Arch Otorhinolaryngol. 2024. PMID: 38308762
-
Nivolumab-induced autoimmune diabetes mellitus presenting as diabetic ketoacidosis in a patient with metastatic lung cancer.J Immunother Cancer. 2017 May 16;5:40. doi: 10.1186/s40425-017-0245-2. eCollection 2017. J Immunother Cancer. 2017. PMID: 28515940 Free PMC article.
-
Immune checkpoint inhibitor-induced refractory polyarthritis rapidly improved by sarilumab and monitoring with joint ultrasonography: A case report.Medicine (Baltimore). 2022 Jan 14;101(2):e28428. doi: 10.1097/MD.0000000000028428. Medicine (Baltimore). 2022. PMID: 35029182 Free PMC article.
-
Nivolumab-induced diabetes mellitus-a case report with literature review of the treatment options.Front Immunol. 2023 Oct 26;14:1248919. doi: 10.3389/fimmu.2023.1248919. eCollection 2023. Front Immunol. 2023. PMID: 37965350 Free PMC article. Review.
-
Immune Checkpoint Inhibitor-Induced Pure Red Cell Aplasia: A Review of 2 Cases in Metastatic Melanoma.Am J Case Rep. 2023 Nov 14;24:e941789. doi: 10.12659/AJCR.941789. Am J Case Rep. 2023. PMID: 37957950 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

