Long COVID-19 autoantibodies and their potential effect on fertility
- PMID: 40496870
- PMCID: PMC12149208
- DOI: 10.3389/fimmu.2025.1540341
Long COVID-19 autoantibodies and their potential effect on fertility
Abstract
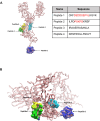
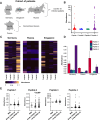
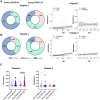
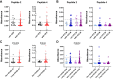
Impaired spermatogenesis has been reported in coronavirus disease 2019 (COVID-19) patients. However, the impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on male fertility remains unclear. The purpose of this multicenter study was to investigate the possible impact of SARS-CoV-2 infection on male fertility and determine the potential reasons leading to impaired male reproductive functions. In silico approach identified ~60 amino acid sequences containing at least five continuous residues shared by SARS-CoV-2 Spike glycoprotein and spermatogenesis-linked proteins. Four synthetic peptides were tested with sera from independent cohorts of patients with acute and long COVID-19 syndrome (LCS), and naïve vaccinated subjects. Immunogenicity and pathogenicity studies were performed by immunizing mice with two selected peptides and testing the antigenicity of induced antibodies. While none of four peptides were recognized by antibodies from vaccinated people, infected patients exhibited high reactivity to peptide 4, and LCS patients, especially women, showed elevated antibody levels against peptide 2. Women with LCS and chronic fatigue syndrome had higher levels of peptide 2-reacting antibodies than those with idiopathic chronic fatigue syndrome. Noteworthy, peptide 2 antibodies showed, in in vitro experiment, a specific interaction with mouse testicular tissue antigens. These findings raise the possibility that cross-reactive epitopes between SARS-CoV-2 Spike protein and spermatogenesis-related antigens may affect infected patients' fertility, suggesting a potential for autoimmune responses with human consequences.
Keywords: autoantibodies; coronavirus infection; male reproductive system; peptide sequence identity; post-COVID-19 condition.
Copyright © 2025 Talamini, Fonseca, Kanduc, Chaloin, Verdot, Galmiche, Dotan, Filgueiras, Borghi, Meroni, Gavrilova, Ryabkova, Churilov, Halpert, Lensch, Thurner, Fong, Ng, Rénia, Young, Lye, Lozano, Cabral-Marques, Shoenfeld and Muller.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

