Distinct Types of Regulated Cell Death in Melanoma
- PMID: 40497999
- PMCID: PMC12154313
- DOI: 10.3390/cells14110823
Distinct Types of Regulated Cell Death in Melanoma
Abstract
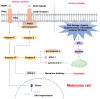
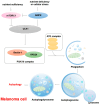
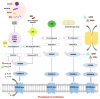
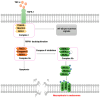
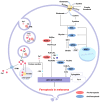
Resistance to cell death is one of the core hallmarks of cancer, with regulatory abnormalities particularly pronounced in the malignant progression and therapeutic resistance of melanoma. This review aims to systematically summarize the roles and mechanisms of regulated cell death (RCD) in melanoma. Currently, distinct types of RCD, including apoptosis, autophagy, pyroptosis, immunogenic cell death, necroptosis, and ferroptosis, have all been found to be involved in melanoma. Autophagy promotes the survival of melanoma cells under stress conditions through metabolic adaptation, yet its excessive activation can trigger cell death. Immunogenic cell death has the capacity to elicit adaptive immune responses in immunocompetent syngeneic hosts. Necroptosis, governed by the receptor-interacting protein kinase 1 (RIPK1)/RIPK3 mixed lineage kinase domain-like protein (MLKL) signaling axis, can synergize with immunotherapy to enhance anti-melanoma immune responses when activated. Pyroptosis, mediated by Gasdermin proteins, induces the release of inflammatory factors that reshape the tumor microenvironment and enhance the efficacy of immune checkpoint inhibitors. Ferroptosis, characterized by lipid peroxidation, can overcome melanoma resistance by targeting the solute carrier family 7 member 11 (SLC7A11)/glutathione peroxidase 4 (GPX4) axis. Therapeutic strategies targeting RCD pathways have demonstrated breakthrough potential. Several agents have been developed to target RCD in order to suppress melanoma.
Keywords: apoptosis; autophagy; ferroptosis; immunogenic cell death; melanoma; necroptosis; pyroptosis; regulatory cell death.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Regulated Necrotic Cell Death in Alternative Tumor Therapeutic Strategies.Cells. 2020 Dec 17;9(12):2709. doi: 10.3390/cells9122709. Cells. 2020. PMID: 33348858 Free PMC article. Review.
-
Targeting cell death pathways for cancer therapy: recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research.J Hematol Oncol. 2022 Dec 8;15(1):174. doi: 10.1186/s13045-022-01392-3. J Hematol Oncol. 2022. PMID: 36482419 Free PMC article. Review.
-
Heme Oxgenase-1, a Cardinal Modulator of Regulated Cell Death and Inflammation.Cells. 2021 Feb 28;10(3):515. doi: 10.3390/cells10030515. Cells. 2021. PMID: 33671004 Free PMC article. Review.
-
A comprehensive investigation of associations between cell death pathways and molecular and clinical features in pan-cancer.Clin Transl Oncol. 2025 Jun;27(6):2731-2749. doi: 10.1007/s12094-024-03769-x. Epub 2024 Nov 2. Clin Transl Oncol. 2025. PMID: 39487950
-
Targeting novel regulated cell death: Ferroptosis, pyroptosis and necroptosis in anti-PD-1/PD-L1 cancer immunotherapy.Cell Prolif. 2024 Aug;57(8):e13644. doi: 10.1111/cpr.13644. Epub 2024 Apr 9. Cell Prolif. 2024. PMID: 38594879 Free PMC article. Review.
References
-
- Garbe C., Amaral T., Peris K., Hauschild A., Arenberger P., Basset-Seguin N., Bastholt L., Bataille V., Del Marmol V., Dréno B., et al. European consensus-based interdisciplinary guideline for melanoma. Part 1: Diagnostics: Update 2022. Eur. J. Cancer. 2022;170:236–255. doi: 10.1016/j.ejca.2022.03.008. - DOI - PubMed
-
- Tribble J.T., Brownell I., Cahoon E.K., Sargen M.R., Shiels M.S., Engels E.A., Volesky-Avellaneda K.D. A Comparative Study of Merkel Cell Carcinoma and Melanoma Incidence and Survival in the United States, 2000–2021. J. Investig. Dermatol. 2025 doi: 10.1016/j.jid.2024.12.014. in press . - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

