Deletion of PPARα in mouse brown adipocytes increases their De Novo Lipogenesis
- PMID: 40499650
- PMCID: PMC12221382
- DOI: 10.1016/j.molmet.2025.102184
Deletion of PPARα in mouse brown adipocytes increases their De Novo Lipogenesis
Abstract
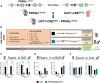
Objective: Peroxisome Proliferator-Activated Receptors (PPARs) are nuclear receptors involved in the control of lipid metabolism. The PPARα isoform is highly expressed in brown adipose tissue (BAT). However, its precise role in BAT remains unclear. Here, we aimed to investigate the role of PPARα in BAT of high fat diet-induced obese mice in a thermoneutral environment.
Methods: We used tamoxifen-inducible-BAT specific PPARα knockout mice (PPARαBATKO) that were housed at thermoneutrality to minimize BAT basal activation, fed a high-fat diet for 20 weeks and challenged with a β3-adrenergic agonist (CL316,243) during the last week. Both male and female mice were studied.
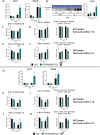
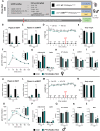
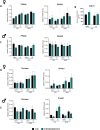
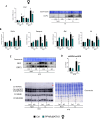
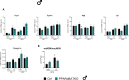
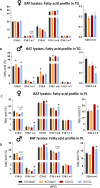
Results: Body weight and glucose tolerance tests were similar in both sexes and genotypes. However, BAT morphology was altered in PPARαBATKO mice, with more unilocular and larger lipid droplets compared to control mice, suggesting BAT impaired function. Indeed, when treated with CL316,243, both male and female mice had increased De Novo Lipogenesis (DNL), reflected by an increased expression of ChREBPβ and lipogenic enzymes ACLY, ACC1, FASN and SCD1. These changes were accompanied by an increase in fatty acids in triglycerides, and thus an increase in lipid storage. Moreover, lipid profiles in phospholipids were different, suggesting a modification in the membrane content with an increase of palmitoleate.
Conclusions: Altogether, our results reveal a key role for PPARα in DNL in BAT and in the regulation of lipid metabolism in HFD-induced obesity.
Keywords: Brown adipose tissue; High fat diet-induced obesity; Inducible UCP1-CRE; Lipid metabolism; Peroxisome proliferator-activated receptor alpha; β(3)-adrenergic stimulation.
Copyright © 2025 The Author(s). Published by Elsevier GmbH.. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






References
-
- Collaborators G.B.D.O., Afshin A., Forouzanfar M.H., Reitsma M.B., Sur P., Estep K., et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017;377(1):13–27. https://10.1056/NEJMoa1614362 - DOI - PMC - PubMed
-
- Collaboration N.C.D.R.F. Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403(10431):1027–1050. https://10.1016/S0140-6736(23)02750-2 - DOI - PMC - PubMed
-
- Virtanen K.A., Lidell M.E., Orava J., Heglind M., Westergren R., Niemi T., et al. Functional brown adipose tissue in healthy adults. N Engl J Med. 2009;360(15):1518–1525. https://10.1056/NEJMoa0808949 - DOI - PubMed
-
- van Marken Lichtenbelt W.D., Vanhommerig J.W., Smulders N.M., Drossaerts J.M., Kemerink G.J., Bouvy N.D., et al. Cold-activated brown adipose tissue in healthy men. N Engl J Med. 2009;360(15):1500–1508. https://10.1056/NEJMoa0808718 - DOI - PubMed
-
- Cypess A.M., Lehman S., Williams G., Tal I., Rodman D., Goldfine A.B., et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med. 2009;360(15):1509–1517. https://10.1056/NEJMoa0810780 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

