Autophagy-enhancing strategies to promote intestinal viral resistance and mucosal barrier function in SARS-CoV-2 infection
- PMID: 40503169
- PMCID: PMC12153388
- DOI: 10.1080/27694127.2025.2514232
Autophagy-enhancing strategies to promote intestinal viral resistance and mucosal barrier function in SARS-CoV-2 infection
Abstract
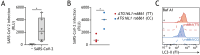
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative agent of Coronavirus disease 19 (COVID-19), continues to circulate globally despite the widespread vaccination and therapeutics like Paxlovid, remdesivir, and molnupiravir. COVID-19 is associated with both respiratory and gastrointestinal manifestations, with persistent intestinal pathology contributing to the post-COVID-19 condition. We have previously demonstrated the antiviral activity of autophagy-blocking drugs, such as Berbamine dihydrochloride, against intestinal SARS-CoV-2 acquisition. In addition, the autophagy blockers restored the barrier function of infected intestinal epithelium. In this addendum, using human intestinal organoids, we present evidence for a protective role of intrinsic higher levels of autophagy flux in limiting intestinal SARS-CoV-2 infection. Pharmacological treatment with Akt inhibitor MK-2206 hydrochloride suppressed viral entry into the intestinal epithelium. This antiviral effect of MK-2206 was shown to be dependent on Synaptosomal-associated protein 29-dependent (SNAP-29)-mediated autophagy flux. Furthermore, extrinsically enhanced autophagy with MK-2206 also prevented SARS-CoV-2-induced intestinal barrier damage. Our findings thus underscore the intricate role of autophagy pathways in the dissemination and pathogenesis of intestinal SARS-CoV-2, highlighting the therapeutic potential of host-directed therapies targeting autophagy to intervene in COVID-19-associated sequelae and improve intestinal health.
Keywords: Antiviral immunity; SARS-CoV-2; autophagy; epithelial cells; gastrointestinal pathology; host-directed therapy; human intestinal organoids; post-COVID-19 condition.
© 2025 The Author(s). Published by Informa UK Limited, trading as Taylor & Francis Group.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures


References
-
- WHO . Coronavirus disease (COVID-19). 2023. https://www.who.int/health-topics/coronavirus
-
- Mathieu E, Ritchie H, Rodés-Guirao L, et al. COVID-19 pandemic 2020. Available from: https://ourworldindata.org/coronavirus
-
- WHO . Post COVID-19 condition (long COVID). 2025. https://www.who.int/news-room/fact-sheets/detail/post-covid-19-condition...)
LinkOut - more resources
Full Text Sources
Miscellaneous
