Human amyotrophic lateral sclerosis/motor neuron disease: The disease-associated microglial pathway is upregulated while APOE genotype governs risk and survival
- PMID: 40506843
- PMCID: PMC12488259
- DOI: 10.1111/bpa.70019
Human amyotrophic lateral sclerosis/motor neuron disease: The disease-associated microglial pathway is upregulated while APOE genotype governs risk and survival
Abstract
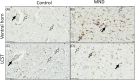
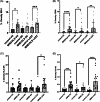
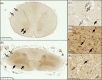
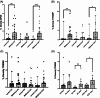
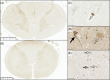
A key role for inflammation in amyotrophic lateral sclerosis/motor neuron disease (ALS/MND) has been identified. It is vital to assess which central nervous system structures are most affected and which inflammatory processes are responsible in humans. The inflammatory transcriptome was characterized in the cervical spinal cord and motor cortex in post-mortem frozen and formalin-fixed paraffin-embedded specimens from human sporadic ALS/MND and control cases using the nCounter® Neuroinflammation Panel. Archival data were reanalyzed and compared with the nCounter data. Immunohistochemistry was used to examine the inflammatory response in the spinal cord and motor cortex and validate changes found during transcriptomic analyses. In the spinal cord, marked inflammation was observed, while less inflammation was detected in the motor cortex. Examination of differentially expressed genes in the spinal cord highlighted TREM2, TYROBP, APOE, and CD163, as well as phagocytic pathways. In sporadic ALS/MND spinal cord, significant microglial reactivity and involvement of TREM2, ApoE (encoded by APOE), and TYROBP were confirmed, suggesting the involvement of the disease-associated microglial (DAM) phenotype. The corticospinal tracts showed greater inflammation than the ventral horns. The precentral gyrus of ALS/MND again showed less immune reactivity to disease when compared to controls. Finally, in the largest cohort assessed to date, we demonstrate an association between the APOE variant and ALS/MND risk, age of onset, and survival. We find confirmed associations between APOE ε3/ε3 and disease and between ε2/ε2 and absence of disease. Further, ε4/ε4 appears to be associated with earlier disease onset and a more aggressive course. We conclude that while there is widespread inflammation in the CNS in sporadic ALS/MND, this is more marked in the spinal cord, especially the corticospinal tract. The specific markers stress the DAM phenotype as having a key role together with a possible influx of somatic macrophages. In addition, APOE function and genotype may be relevant in ALS/MND.
Keywords: APOE; amyotrophic lateral sclerosis; disease‐associated microglia; inflammation; motor neuron disease.
© 2025 The Author(s). Brain Pathology published by John Wiley & Sons Ltd on behalf of International Society of Neuropathology.
Conflict of interest statement
None.
Figures




References
-
- Ince PG, Highley JR, Wharton SB. Motor neuron disorders. In: Love S, Perry A, Ironside J, Budka H, editors. Greenfield's neuropathology. Abingdon, Oxon, UK: Taylor & Francis; 2015. p. 817–848.
-
- Herculano‐Houzel S. The glia/neuron ratio: how it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia. 2014;62(9):1377–1391. - PubMed
-
- Mittelbronn M, Dietz K, Schluesener HJ, Meyermann R. Local distribution of microglia in the normal adult human central nervous system differs by up to one order of magnitude. Acta Neuropathol. 2001;101(3):249–255. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

