TSGA10 as a Model of a Thermal Metabolic Regulator: Implications for Cancer Biology
- PMID: 40507237
- PMCID: PMC12153768
- DOI: 10.3390/cancers17111756
TSGA10 as a Model of a Thermal Metabolic Regulator: Implications for Cancer Biology
Abstract
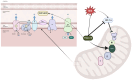
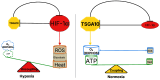
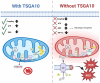
TSGA10, a multifunctional protein critical for mitochondrial coupling and metabolic regulation, plays a paradoxical role in cancer progression and carcinogenesis. Here, we outline a potential mechanism by which TSGA10 mediates metabolism in oncogenesis and thermal modulation. Initially identified in spermatogenesis, TSGA10 interacts with mitochondrial Complex III: it directly binds cytochrome c1 (CytC1). In our model, TSGA10 optimizes electron transport to minimize reactive oxygen species (ROS) and heat production while enhancing Adenosine Triphosphate (ATP) synthesis. In cancer, TSGA10's expression is context-dependent: Its downregulation in tumors like glioblastoma might disrupt mitochondrial coupling, promoting electron leakage, ROS accumulation, and genomic instability. This dysfunction would be predicted to contribute to a glycolytic shift, facilitating tumor survival under hypoxia. Conversely, TSGA10 overexpression in certain cancers suppresses HIF-1α, inhibiting glycolysis and metastasis. TSGA10 and HIF-1α engage in mutual counter-regulation-TSGA10 represses HIF-1α to sustain oxidative phosphorylation (OXPHOS), while HIF-1α suppression of TSGA10 under hypoxia or thermal stress amplifies glycolytic dependency. This interplay is pivotal in tumors adapting to microenvironmental stressors, such as cold-induced mitochondrial uncoupling, which mimics brown adipose tissue thermogenesis to reduce ROS and sustain proliferation. Tissue-specific TSGA10 expression further modulates cancer susceptibility: high levels in the testes and brain may protect against thermal and oxidative damage, whereas low expression in the liver permits HIF-1α-driven metabolic plasticity. Altogether, our model suggests that TSGA10 plays a central role in mitochondrial fidelity. We suggest that its crosstalk with oncogenic pathways position it as a metabolic rheostat, whose dysregulation fosters tumorigenesis through ROS-mediated mutagenesis, metabolic reprogramming, and microenvironmental remodeling. Targeting the hypothesized TSGA10-mediated mitochondrial coupling may offer therapeutic potential to disrupt cancer's adaptive energetics and restore metabolic homeostasis.
Keywords: Complex III (CytC1); HIF-1α; TSGA10; Warburg effect; carcinogenesis; mitochondrial coupling.
Conflict of interest statement
The authors declare that there are no potential commercial or financial conflicts of interest that could be perceived as influencing the research reported in this manuscript. The authors affiliated with MDPI confirm that their participation in the study was conducted in an academic and scientific capacity, independent of any commercial interests. All data analysis, interpretation, and conclusions were carried out without bias and in adherence to the ethical guidelines set forth by MDPI.
Figures

Similar articles
-
TSGA10 Over Expression Decreases Metastasic and Metabolic Activity by Inhibiting HIF-1 in Breast Cancer Cells.Arch Med Res. 2020 Jan;51(1):41-53. doi: 10.1016/j.arcmed.2019.12.002. Epub 2020 Feb 18. Arch Med Res. 2020. PMID: 32086108
-
Tumor microenvironment and metabolic synergy in breast cancers: critical importance of mitochondrial fuels and function.Semin Oncol. 2014 Apr;41(2):195-216. doi: 10.1053/j.seminoncol.2014.03.002. Epub 2014 Mar 5. Semin Oncol. 2014. PMID: 24787293 Review.
-
New function of TSGA10 gene in angiogenesis and tumor metastasis: a response to a challengeable paradox.Hum Mol Genet. 2016 Jan 15;25(2):233-44. doi: 10.1093/hmg/ddv461. Epub 2015 Nov 15. Hum Mol Genet. 2016. PMID: 26573430
-
TSGA10 overexpression inhibits angiogenesis of HUVECs: A HIF-2α biased perspective.Microvasc Res. 2020 Mar;128:103952. doi: 10.1016/j.mvr.2019.103952. Epub 2019 Nov 5. Microvasc Res. 2020. PMID: 31704243
-
HIF-1alpha modulates energy metabolism in cancer cells by inducing over-expression of specific glycolytic isoforms.Mini Rev Med Chem. 2009 Aug;9(9):1084-101. doi: 10.2174/138955709788922610. Mini Rev Med Chem. 2009. PMID: 19689405 Review.
References
-
- Salehipour P., Nematzadeh M., Mobasheri M.B., Afsharpad M., Mansouri K., Modarressi M.H. Identification of new TSGA10 transcript variants in human testis with conserved regulatory RNA elements in 5’untranslated region and distinct expression in breast cancer. Biochim. Biophys. Acta (BBA) Gene Regul. Mech. 2017;1860:973–982. doi: 10.1016/j.bbagrm.2017.07.007. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

