Mechanistic Insights into Anti-Nectin4-VcMMAE-Induced Ocular Toxicity: From Cellular Uptake Pathways to Molecular Modification
- PMID: 40507807
- PMCID: PMC12154390
- DOI: 10.3390/ijms26114996
Mechanistic Insights into Anti-Nectin4-VcMMAE-Induced Ocular Toxicity: From Cellular Uptake Pathways to Molecular Modification
Abstract
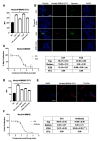
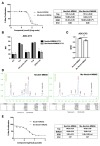
Antibody-drug conjugates (ADCs) represent a novel approach to cancer treatment. Enfortumab vedotin (PADCEV), as a prominent example, has demonstrated remarkable clinical efficacy. However, its ocular toxicity has raised concerns. This study aimed to explore the molecular mechanisms underlying PADCEV-induced ocular toxicity. SD rats, whose ocular structures are similar to those of humans, were selected to establish an ocular toxicity model to mimic the human response. In vitro experiments were conducted using human primary corneal epithelial cells, HCE-T. The results confirmed that nectin-4 plays a crucial role in the cellular uptake of PADCEV, and non-specific pinocytosis is also involved. Additionally, a variant was obtained by introducing point mutations in the Fc region of PADCEV, which was found to reduce corneal epithelial toxicity. The findings of this study not only deepen our understanding of ADC-induced ocular toxicity but also provide new insights into optimizing ADC design and enhancing treatment safety.
Keywords: endocytosis mechanism; enfortumab vedotin; molecular optimization; ocular toxicity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Nectin-4-directed antibody-drug conjugates (ADCs): Spotlight on preclinical and clinical evidence.Life Sci. 2024 Sep 1;352:122910. doi: 10.1016/j.lfs.2024.122910. Epub 2024 Jul 11. Life Sci. 2024. PMID: 39002610 Review.
-
Biomarkers of Response to Anti-NECTIN4 Antibody-Drug Conjugate Enfortumab Vedotin in Urothelial Cancer.Eur Urol Focus. 2024 Mar;10(2):224-226. doi: 10.1016/j.euf.2024.04.001. Epub 2024 Apr 16. Eur Urol Focus. 2024. PMID: 38631991 Review.
-
Modulation of Macropinocytosis-Mediated Internalization Decreases Ocular Toxicity of Antibody-Drug Conjugates.Cancer Res. 2018 Apr 15;78(8):2115-2126. doi: 10.1158/0008-5472.CAN-17-3202. Epub 2018 Jan 30. Cancer Res. 2018. PMID: 29382707
-
Enfortumab Vedotin-ejfv: A First-in-Class Anti-Nectin-4 Antibody-Drug Conjugate for the Management of Urothelial Carcinoma.Ann Pharmacother. 2021 Jun;55(6):772-782. doi: 10.1177/1060028020960402. Epub 2020 Sep 18. Ann Pharmacother. 2021. PMID: 32945172
-
Exploring membranous NECTIN-4 expression patterns and enfortumab vedotin response in prostate cancer.J Cell Mol Med. 2024 Jul;28(14):e18572. doi: 10.1111/jcmm.18572. J Cell Mol Med. 2024. PMID: 39072867 Free PMC article.
References
-
- Moolten F.L., Capparell N.J., Cooperband S.R. Antitumor effects of antibody-diphtheria toxin conjugates: Use of hapten-coated tumor cells as an antigenic target. J. Natl. Cancer Inst. 1972;49:1057–1062. - PubMed
-
- Beck A., Reichert J.M. Antibody-Drug Conjugates. Proc. Natl. Acad. Sci. USA. 2013;6:15–17. doi: 10.4161/mabs.27436. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

