Targeting of Epithelial Cell Adhesion Molecule-Expressing Malignant Tumors Using an Albumin-Binding Domain-Fused Designed Ankyrin Repeat Protein: Effect of the Molecular Architecture
- PMID: 40508045
- PMCID: PMC12154114
- DOI: 10.3390/ijms26115236
Targeting of Epithelial Cell Adhesion Molecule-Expressing Malignant Tumors Using an Albumin-Binding Domain-Fused Designed Ankyrin Repeat Protein: Effect of the Molecular Architecture
Abstract
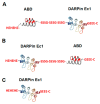
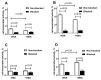
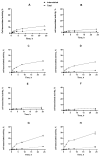
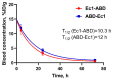
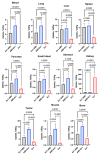
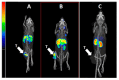
Designed ankyrin repeat protein (DARPin) Ec1, a small scaffold protein (18 kDa), binds with high affinity the epithelial cell adhesion molecule (EpCAM) that is overexpressed in several carcinomas. To enhance the targeted delivery of cytotoxic drugs using Ec1, we investigated the potential of fusing Ec1 with an albumin-binding domain (ABD) to improve its circulation time and decrease renal uptake. Two fusion proteins were created, Ec1-ABD, with the ABD at the C-terminus, and ABD-Ec1, with the ABD at the N-terminus. Both variants were labeled with 111In. ABD-fused variants bound specifically to EpCAM-expressing cells with picomolar affinity. Adding human albumin reduced the affinity. This effect was more pronounced for Ec1-ABD; however, the affinity remained in the subnanomolar range. The position of the ABD did not influence the internalization rate of both variants by human cancer cells. In mouse models with human cancer xenografts, both variants demonstrated over 10-fold lower renal uptake compared to the Ec1. Tumor uptake of the ABD-fused variants was higher than the uptake of Ec1. ABD-Ec1 provided two-fold higher tumor uptake, indicating fusion with an ABD as a promising way to modulate the targeting properties of an Ec1-based construct. However, the effect of fusion depends on the order of the domains.
Keywords: albumin-binding domain (ABD); designed ankyrin repeat protein; epithelial cell adhesion molecule (EpCAM); fusion protein; nude mice; xenograft.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
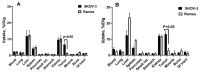
Similar articles
-
Designed Ankyrin Repeat Protein-Mediated Peptide Nucleic Acid-Based Pretargeting: A Proof-of-Principle Study.J Nucl Med. 2025 Jul 1;66(7):1105-1111. doi: 10.2967/jnumed.125.269533. J Nucl Med. 2025. PMID: 40374553
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
GPA33-pretargeted radioimmunotherapy with mono- and bivalent DOTA-based Lu-177-labeled radiohaptens in a mouse orthotopic liver xenograft model of metastatic human colorectal cancer.Theranostics. 2025 May 25;15(13):6274-6289. doi: 10.7150/thno.107209. eCollection 2025. Theranostics. 2025. PMID: 40521192 Free PMC article.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
References
-
- Colombo R., Tarantino P., Rich J.R., LoRusso P.M., de Vries E.G.E. The Journey of Antibody-Drug Conjugates: Lessons Learned from 40 Years of Development. Cancer Discov. 2024;14:2089–2108. doi: 10.1158/2159-8290.CD-24-0708. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

