Niacin Modulates SIRT1-Driven Signaling to Counteract Radiation-Induced Neurocognitive and Behavioral Impairments
- PMID: 40508105
- PMCID: PMC12155517
- DOI: 10.3390/ijms26115285
Niacin Modulates SIRT1-Driven Signaling to Counteract Radiation-Induced Neurocognitive and Behavioral Impairments
Abstract
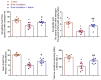
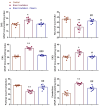
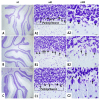
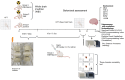
Radiation exposure causes neuroinflammation, oxidative stress, and neuronal loss, leading to cognitive and behavioral impairments. This study aims to evaluate the effect of niacin interventions on whole-brain irradiation (WBI)-induced cognitive and behavioral impairment. Female Wistar rats were randomly assigned to Control (Group 1), Radiation +Saline (Group 2), and Radiation +niacin (Group 3) groups. Rats in the irradiated groups (Groups 2 and 3) received a single dose of 20 Gy photon irradiation. Group 2 received water seven days after irradiation, while Group 3 received niacin (60 mg/kg, 2 mL) oral gavage for 15 days. On days 22, 23, and 24, behavioral assessments were performed, including the Open Field Test, the Sociability Test, and the Passive Avoidance Learning (PAL) task. Biochemical analyses included MDA, BDNF, TNF-α, CREB), SIRT1, and SIRT6 measured by ELISA. Histological assessments included neuronal density and GFAP immunostaining in CA1 and CA3 regions of the hippocampus and cerebellar Purkinje neurons. Radiation exposure importantly increased MDA and TNF-α levels, while SIRT1, SIRT6, BDNF, and CREB were notably reduced. This was accompanied by neuronal loss in the cerebellum and hippocampus, astrogliosis, and behavioral and cognitive deficits. Niacin treatment significantly decreased MDA and TNF-α levels while increasing BDNF, CREB, SIRT1, and SIRT6 expression, attenuating neuronal apoptosis. Immunohistochemical analysis demonstrated that niacin treatment enhanced neuronal density in the CA1 and CA3 regions of the hippocampus and cerebellar Purkinje neurons while reducing GFAP immunoreactivity in the CA1, CA3, and cerebellum following WBI. Behaviorally, niacin treatment improved social interaction, locomotor activity, and memory performance, underscoring its neuroprotective potential against WBI-induced damage. These findings suggest that niacin may ameliorate behavioral and cognitive impairments following whole brain irradiation by activating the SIRT1/CREB/BDNF or SIRT1/SIRT6/MDA/TNF-α signaling pathway.
Keywords: SIRT1; SIRT6; cognitive function; neuroinflammation; neuroprotection; niacin; oxidative stress; whole brain irradiation.
Conflict of interest statement
The authors report no conflicts of interest.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

