Adaptive hydrogel loaded with pre-coordinated stem cells for enhanced osteoarthritis therapy
- PMID: 40510751
- PMCID: PMC12159505
- DOI: 10.1016/j.bioactmat.2025.05.018
Adaptive hydrogel loaded with pre-coordinated stem cells for enhanced osteoarthritis therapy
Abstract
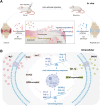
Osteoarthritis (OA) is a prevalent chronic joint disease with no currently available cure. Despite the promise of mesenchymal stromal cells (MSCs) in promoting OA management, direct intra-articular administration of MSCs faces several critical challenges, including rapid cell clearance from the joint cavity, limited survival in the hostile inflammatory environment, and insufficient control over their differentiation. In this study, we present a strategy that enhances the functionality of MSCs via pre-coordinated with Mg2+ and hypoxia-mimicking agent dimethyloxalylglycine (DMOG) integrated within an adaptive hydrogel for OA treatment. Mg2+ regulates macrophage polarization toward an anti-inflammatory phenotype, inhibits osteoclast activation, and preserves subchondral bone integrity by activating the PI3K-Akt signaling pathway. Concurrently, DMOG, activates the HIF-1α pathway, mimicking hypoxic microenvironment that support chondrocyte repair and stimulate cartilage matrix synthesis. MSCs pre-coordinated with Mg2+ and DMOG exhibit enhanced chondrogenic differentiation and immunomodulatory capacity, thus improving their regenerative potential in OA. To facilitate localized and sustained delivery, a self-healing tissue-adhesive hydrogel composed of phenylboronic acid and methacrylate-modified hyaluronic acid (HAMA-PBA) is synthesized to encapsulate the pre-coordinated MSCs. This hydrogel ensures cellular retention and functionality at the injury site. In vivo, the system significantly reduces joint inflammation, enhances cartilage regeneration, and improves joint function. Overall, these findings demonstrate a synergistic and effective stem cell-based therapeutic strategy for OA treatment through biochemical preconditioning and biomaterial-enabled delivery.
Keywords: Cell therapy; Chondrogenic; Hypoxia; Injectable hydrogel; Osteoarthritis.
© 2025 The Authors.
Conflict of interest statement
Qing Cai is an editorial board member for Bioactive Materials and was not involved in the editorial review or decision to publish this article. All authors declare that they have no competing interests.
Figures










References
-
- Xie Y., Zinkle A., Chen L., Mohammadi M. Fibroblast growth factor signalling in osteoarthritis and cartilage repair. Nat. Rev. Rheumatol. 2020;16:547–564. - PubMed
-
- Cho Y., Kim H., Yook G., Yong S., Kim S., Lee N., Kim Y.J., Kim J.H., Kim T.W., Chang M.J., Lee K.M., Chang C.B., Kang S.B., Kim J.H. Predisposal of interferon regulatory factor 1 deficiency to accumulate DNA damage and promote osteoarthritis development in cartilage. Arthritis Rheumatol. 2024;76:882–893. - PubMed
LinkOut - more resources
Full Text Sources

