Lachnospiraceae bacterium alleviates alcohol-associated liver disease by enhancing N-acetyl-glutamic acid levels and inhibiting ferroptosis through the KEAP1-NRF2 pathway
- PMID: 40511521
- PMCID: PMC12169036
- DOI: 10.1080/19490976.2025.2517821
Lachnospiraceae bacterium alleviates alcohol-associated liver disease by enhancing N-acetyl-glutamic acid levels and inhibiting ferroptosis through the KEAP1-NRF2 pathway
Abstract
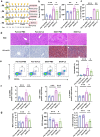
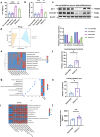
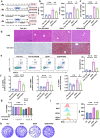
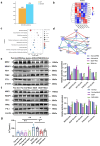
Alcohol-associated liver disease (ALD) is a prevalent global health issue primarily caused by excessive alcohol consumption. Recent studies have highlighted the gut-liver axis's protective role against ALD, mainly through gut microbiota. However, the precise mechanism remains ill-defined. Our results showed a significant reduction in Lachnospiraceae bacterium in the gut microbiota of ALD patients and ethanol (EtOH)-fed mice, as revealed by 16S rDNA sequencing. Supplementation with Lachnospiraceae bacterium strains in mice significantly reduced inflammation, hepatic neutrophil infiltration, oxidative stress, and improved gut microbiota and intestinal permeability. Multi-omics analysis identified N-Acetyl-glutamic acid (NAG) as the most significantly altered metabolite following Lachnospiraceae bacterium supplementation, with levels positively correlated to Lachnospiraceae bacterium colonization. NAG treatment exhibited significant protective effects in EtOH-exposed hepatocyte cell lines and EtOH-fed mice. Mechanistically, NAG confers hepatoprotection against ALD by activating the KEAP1-NRF2 pathway, inhibiting ferroptosis. Notably, the protective effects of NAG were reversed by the NRF2 inhibitor. In conclusion, oral supplementation with Lachnospiraceae bacterium mitigates alcohol-induced liver damage both in vivo and in vitro by inhibiting ferroptosis through NAG-mediated activation of the KEAP1-NRF2 pathway. Lachnospiraceae bacterium may serve as promising probiotics for future clinical applications.
Keywords: Alcohol-associated liver disease; Lachnospiraceae bacterium; N-Acetyl-glutamic acid; NRF2; ferroptosis.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures


Similar articles
-
BNIP3-mediated mitophagy attenuates hypoxic-ischemic brain damage in neonatal rats by inhibiting ferroptosis through P62-KEAP1-NRF2 pathway activation to maintain iron and redox homeostasis.Acta Pharmacol Sin. 2025 Jan;46(1):33-51. doi: 10.1038/s41401-024-01365-x. Epub 2024 Aug 23. Acta Pharmacol Sin. 2025. PMID: 39179868
-
Artemisinin inhibits neuronal ferroptosis in Alzheimer's disease models by targeting KEAP1.Acta Pharmacol Sin. 2025 Feb;46(2):326-337. doi: 10.1038/s41401-024-01378-6. Epub 2024 Sep 9. Acta Pharmacol Sin. 2025. PMID: 39251858
-
Targeting NOTCH1-KEAP1 axis retards chronic liver injury and liver cancer progression via regulating stabilization of NRF2.J Exp Clin Cancer Res. 2025 Aug 9;44(1):232. doi: 10.1186/s13046-025-03488-3. J Exp Clin Cancer Res. 2025. PMID: 40781716 Free PMC article.
-
Ferroptosis and gut microbiota: A new horizon in alcohol-associated liver disease management.Cell Mol Life Sci. 2025 Jul 19;82(1):282. doi: 10.1007/s00018-025-05815-5. Cell Mol Life Sci. 2025. PMID: 40682668 Free PMC article. Review.
-
Synbiotics, prebiotics and probiotics for solid organ transplant recipients.Cochrane Database Syst Rev. 2022 Sep 20;9(9):CD014804. doi: 10.1002/14651858.CD014804.pub2. Cochrane Database Syst Rev. 2022. PMID: 36126902 Free PMC article.
References
-
- Griswold MG, Fullman N, Hawley C, Arian N, Zimsen SRM, Tymeson HD, Venkateswaran V, Tapp AD, Forouzanfar MH, Salama JS, et al. Alcohol use and burden for 195 countries and territories, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2018;392(10152):1015–1035. doi: 10.1016/S0140-6736(18)31310-2. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
