Regulation of redox homeostasis by ATF4-MTHFD2 axis during white adipose tissue browning
- PMID: 40516456
- PMCID: PMC12205661
- DOI: 10.1016/j.redox.2025.103715
Regulation of redox homeostasis by ATF4-MTHFD2 axis during white adipose tissue browning
Abstract
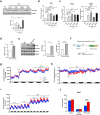
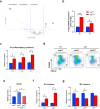
Maintaining redox balance is crucial for mitochondrial homeostasis. During browning of white adipocytes, both the quality and quantity of mitochondria undergo dramatic changes. However, the mechanisms controlling the redox balance in the mitochondria during this process remain unclear. In this study, we demonstrate that thermogenic activation occurs before mitochondrial biogenesis during cold-induced browning of inguinal white adipose tissue (iWAT) and is accompanied by increased mitochondrial stress and integrated stress response (ISR) signaling. Specifically, cold exposure enhances the expression of ATF4, an ISR effector. Adipocyte-specific deletion of ATF4 results in increased energy expenditure, but paradoxically leads to a lower core body temperature, and heightened pro-inflammation in iWAT after cold exposure, which is restored by the antioxidant, MitoQ. Mechanistically, ATF4 regulates the redox balance through MTHFD2, an enzyme involved in mitochondrial redox homeostasis by NADPH generation. Cold exposure upregulates MTHFD2 expression in an ATF4-dependent manner, and its inhibition by DS18561882 in vivo leads to impaired cold-induced mitochondrial respiration similar to the effects of ATF4 loss. These findings suggest that ATF4 is essential for redox balance via MTHFD2, thereby affecting tissue homeostasis during iWAT browning.
Copyright © 2025 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





References
-
- Kemp T.S. The origin of mammalian endothermy: a paradigm for the evolution of complex biological structure. Zool. J. Linn. Soc. 2006;147:473–488. doi: 10.1111/j.1096-3642.2006.00226.x. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

