Building blocks for lung regeneration: Stem cells and niches
- PMID: 40518118
- PMCID: PMC12270064
- DOI: 10.1016/j.mocell.2025.100241
Building blocks for lung regeneration: Stem cells and niches
Abstract
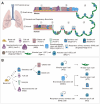
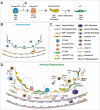
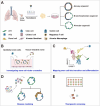
The respiratory system is an essential organ that performs gas exchange through the blood circulation in mammals. Unlike other organs, the lungs are directly exposed to the external environment, including particulate matter, cigarette smoke, and various pollutants, and are therefore, highly susceptible to damage. The lungs retain regional-specific stem/progenitor cells that quickly mobilize to replace the damaged epithelium. Accumulating evidences suggest that fate decision of stem cells relies on regulatory programs integrated by niches constituting the microenvironment providing diverse signals that regulate stem cell behavior. Therefore, understanding cellular diversity and precise interaction between stem cells and their respective niches is crucial to understand how tissue recovers homeostasis after injury. Here, in this review, we summarize recent progress in cellular and functional identity of stem cells and distinctive niches in the lungs. We also describe the molecular mechanism of genetic and epigenetic program in the regulation of stem cell behavior during tissue regeneration. Lastly, we introduce the three-dimensional lung organoid platforms that provide valuable insights into the mechanisms of lung pathophysiology in human system.
Keywords: Alveolar regeneration; Lung organoids; Lung stem cells; Niches; Transitional cell states.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interests The authors have no potential conflicts of interest to disclose.
Figures
Similar articles
-
Interplay between pulmonary epithelial stem cells and innate immune cells contribute to the repair and regeneration of ALI/ARDS.Transl Res. 2024 Oct;272:111-125. doi: 10.1016/j.trsl.2024.05.012. Epub 2024 Jun 17. Transl Res. 2024. PMID: 38897427 Review.
-
Co-colonization and Co-culture of Lung Alveoli Epithelial Stem Cells and Their Endothelial Niche Cells.Methods Mol Biol. 2025;2939:169-181. doi: 10.1007/7651_2025_607. Methods Mol Biol. 2025. PMID: 40106143 Free PMC article.
-
An aberrant immune-epithelial progenitor niche drives viral lung sequelae.Nature. 2024 Oct;634(8035):961-969. doi: 10.1038/s41586-024-07926-8. Epub 2024 Sep 4. Nature. 2024. PMID: 39232171
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Stem-Cell Niches in Health and Disease: Microenvironmental Determinants of Regeneration and Pathology.Cells. 2025 Jun 26;14(13):981. doi: 10.3390/cells14130981. Cells. 2025. PMID: 40643502 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

