Extracellular vesicles isolated from adipose tissue-derived mesenchymal stromal cells as carriers for Paclitaxel delivery
- PMID: 40518531
- PMCID: PMC12168270
- DOI: 10.1186/s13287-025-04435-x
Extracellular vesicles isolated from adipose tissue-derived mesenchymal stromal cells as carriers for Paclitaxel delivery
Abstract
Background: Mesenchymal Stromal Cells (MSC)-derived Extracellular Vesicles (EV) represent innovative tools for drug delivery systems. However, their clinical use is limited by the lack of standardized good manufacturing practice (GMP)-compliant isolation and conservation protocols. In this study, we developed a GMP-compliant protocol for the preparation of MSC-EVs and investigated the feasibility of producing EVs loaded with paclitaxel (PTX) for clinical application as drug products.
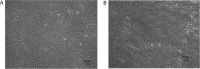
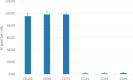
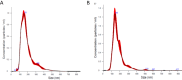
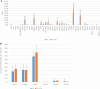
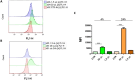
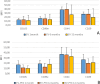
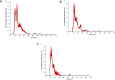
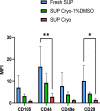
Methods: Adipose tissues from 13 donors were used to obtain MSC-EVs via culture supernatant ultracentrifugation. EVs loaded with PTX were manufactured by adding the drug to the culture medium of MSCs before supernatant collection. EV identity was verified in terms of concentration/size, protein content, morphology, and expression of EV surface markers. The anti-proliferative activity, accumulation ability in tumor cells and PTX content, as well as their stability over time, were also evaluated.
Results: High numbers of EV/EV-PTX compliant in terms of integrity/identity were obtained and can be successfully stored for up to one year at -80 °C. Cellular studies have shown that EVs are capable of accumulating in tumor cells and, when loaded with PTX, inhibiting the proliferation of a pleural mesothelioma cell line.
Conclusions: These results support the potential future clinical use of EVs as carriers for drug delivery to improve cancer treatment strategies.
Keywords: Antitumor drug; Drug delivery systems; Extracellular vesicles; Mesenchymal stromal cells; Paclitaxel.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to partecipate: Samples were collected after signed informed consent of no objection for the use for research of surgical tissues in accordance with the Declaration of Helsinki. The informed consents were obtained prior to tissue collection. The project was approved by ethical committee: Title of the approved project: “human Mesenchymal Stromal Cells (MSCs) loaded with drugs and derived Extracellular Vesicles: production process optimization and drug product characterization”. Name of the institutional approval committee: Institutional Review Board of the IRCCS Neurological Institute C. Besta Foundation. Approval number: 15. Date of approval: March 29, 2023. Human cell lines MSTO-211 H and NCI H2052 were purchased from a commercial vendor, ATCC. The company has confirmed that there was initial ethical approval for the collection of human materials and the derivation of cell lines and that the donors had signed informed consent. See for details: https://www.atcc.org/products/crl-2081 ; https://www.atcc.org/products/crl-5915 . Competing interests: The authors declare that they have no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. The authors declare that they have not use AI-generated work in this manuscript.
Figures



References
-
- Asadi K, Amini A, Gholami A. Mesenchymal stem cell-derived exosomes as a bioinspired nanoscale tool toward nextgeneration cell-free treatment. J Drug Deliv Sci Technol. 2022;77:103856. - DOI

