β-adrenergic signaling modulates breast cancer cell mechanical behaviors through a RhoA-ROCK-myosin II axis
- PMID: 40520106
- PMCID: PMC12164020
- DOI: 10.1016/j.isci.2025.112676
β-adrenergic signaling modulates breast cancer cell mechanical behaviors through a RhoA-ROCK-myosin II axis
Abstract
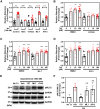
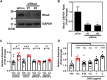
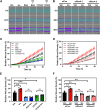
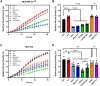
The ability of cancer cells to deform and generate force is implicated in metastasis. We previously showed that β-adrenergic agonists increase cancer cell stiffness, which was associated with enhanced motility and invasion. Here, we investigate how β-adrenoceptor (βAR) activation alters the mechanical behaviors of triple-negative breast cancer cells. We find that βAR activation increases traction forces in metastatic MDA-MB-231HM and MDA-MB-468 cells, but not in non-tumorigenic MCF10A cells. Using computational modeling, we show that βAR activation increases the number of active myosin motors via myosin light chain phosphorylation. To identify molecular regulators, we use a deformability assay to screen for pharmacologic and genetic perturbations. Our results define a βAR-RhoA-ROCK-non-muscle myosin II (NMII) signaling axis that modulates the mechanical behaviors of MDA-MB-231HM and MDA-MB-468 cells. These findings provide insight into how stress signaling regulates cancer cell mechanics and suggest potential targets to block metastasis in triple-negative breast cancer.
Keywords: Cell biology; Functional aspects of cell biology; Mechanobiology.
© 2025 The Author(s).
Conflict of interest statement
The authors declares no competing interests.
Figures

Similar articles
-
Cancer cells become less deformable and more invasive with activation of β-adrenergic signaling.J Cell Sci. 2016 Dec 15;129(24):4563-4575. doi: 10.1242/jcs.194803. Epub 2016 Nov 14. J Cell Sci. 2016. PMID: 27875276 Free PMC article.
-
High extracellular glucose promotes cell motility by modulating cell deformability and contractility via the cAMP-RhoA-ROCK axis in human breast cancer cells.Mol Biol Cell. 2023 Jul 1;34(8):ar79. doi: 10.1091/mbc.E22-12-0560. Epub 2023 May 17. Mol Biol Cell. 2023. PMID: 37195739 Free PMC article.
-
The crosstalk between breast carcinoma-associated fibroblasts and cancer cells promotes RhoA-dependent invasion via IGF-1 and PAI-1.Oncotarget. 2017 Dec 28;9(12):10375-10387. doi: 10.18632/oncotarget.23735. eCollection 2018 Feb 13. Oncotarget. 2017. PMID: 29535813 Free PMC article.
-
Measuring (biological) materials mechanics with atomic force microscopy. 5. Traction force microscopy (cell traction forces).Microsc Res Tech. 2023 Sep;86(9):1069-1078. doi: 10.1002/jemt.24368. Epub 2023 Jun 22. Microsc Res Tech. 2023. PMID: 37345422 Free PMC article. Review.
-
Myosin Light Chains in the Progression of Cancer.Cells. 2024 Dec 17;13(24):2081. doi: 10.3390/cells13242081. Cells. 2024. PMID: 39768172 Free PMC article. Review.
References
-
- Mekhdjian A.H., Kai F., Rubashkin M.G., Prahl L.S., Przybyla L.M., McGregor A.L., Bell E.S., Barnes J.M., DuFort C.C., Ou G., et al. Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix. Mol. Biol. Cell. 2017;28:1467–1488. doi: 10.1091/mbc.E16-09-0654. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

