ALS-associated TDP-43 aggregates drive innate and adaptive immune cell activation
- PMID: 40520109
- PMCID: PMC12167492
- DOI: 10.1016/j.isci.2025.112648
ALS-associated TDP-43 aggregates drive innate and adaptive immune cell activation
Abstract
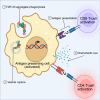
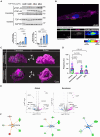
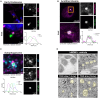
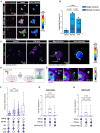
Amyotrophic lateral sclerosis (ALS) is the most common and fatal motor neuron disease. Approximately 90% of ALS patients exhibit pathology of the master RNA regulator, transactive response DNA binding protein (TDP-43). Despite the prevalence TDP-43 pathology in ALS motor neurons, recent findings suggest immune dysfunction is a determinant of disease progression in patients. Whether TDP-43 aggregates elicit immune responses remains underexplored. In this study, we demonstrate that TDP-43 aggregates are internalized by antigen-presenting cell populations, cause vesicle rupture, and drive innate and adaptive immune cell activation by way of antigen presentation. Using a multiplex imaging platform, we observed enrichment of activated microglia/macrophages in ALS white matter that correlated with phosphorylated TDP-43 accumulation, CD8 T cell infiltration, and major histocompatibility complex expression. Taken together, this study sheds light on a novel cellular response to TDP-43 aggregates through an immunological lens.
Keywords: Immunity; Neuroscience; Omics.
© 2025 The Authors.
Conflict of interest statement
The authors declare no competing conflicts of interest.
Figures


Similar articles
-
TDP-43 pathology links innate and adaptive immunity in amyotrophic lateral sclerosis.bioRxiv [Preprint]. 2024 Jan 10:2024.01.07.574541. doi: 10.1101/2024.01.07.574541. bioRxiv. 2024. PMID: 38260395 Free PMC article. Preprint.
-
CK1δ/ε-mediated TDP-43 phosphorylation contributes to early motor neuron disease toxicity in amyotrophic lateral sclerosis.Acta Neuropathol Commun. 2024 Dec 4;12(1):187. doi: 10.1186/s40478-024-01902-z. Acta Neuropathol Commun. 2024. PMID: 39633494 Free PMC article.
-
Deep learning analyses of splicing variants identify the link of PCP4 with amyotrophic lateral sclerosis.Brain. 2025 Jul 7;148(7):2331-2347. doi: 10.1093/brain/awaf025. Brain. 2025. PMID: 39852553
-
TDP-43 as a potential biomarker for amyotrophic lateral sclerosis: a systematic review and meta-analysis.BMC Neurol. 2018 Jun 28;18(1):90. doi: 10.1186/s12883-018-1091-7. BMC Neurol. 2018. PMID: 29954341 Free PMC article.
-
TARDBP-Related Amyotrophic Lateral Sclerosis-Frontotemporal Dementia.2009 Apr 23 [updated 2023 Jan 5]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2009 Apr 23 [updated 2023 Jan 5]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 20301761 Free Books & Documents. Review.
References
-
- Huisman M.H.B., De Jong S.W., Van Doormaal P.T.C., Weinreich S.S., Schelhaas H.J., Van Der Kooi A.J., De Visser M., Veldink J.H., Van Den Berg L.H. Population based epidemiology of amyotrophic lateral sclerosis using capture-recapture methodology. J. Neurol. Neurosurg. Psychiatry. 2011;82:1165–1170. doi: 10.1136/JNNP.2011.244939. - DOI - PubMed
-
- Wijesekera L.C., Mathers S., Talman P., Galtrey C., Parkinson M.H., Ganesalingam J., Willey E., Ampong M.A., Ellis C.M., Shaw C.E., et al. Natural history and clinical features of the flail arm and flail leg ALS variants. Neurology. 2009;72:1087–1094. doi: 10.1212/01.WNL.0000345041.83406.A2. - DOI - PMC - PubMed
-
- Abu-Rumeileh S., Vacchiano V., Zenesini C., Polischi B., de Pasqua S., Fileccia E., Mammana A., Di Stasi V., Capellari S., Salvi F., et al. Diagnostic-prognostic value and electrophysiological correlates of CSF biomarkers of neurodegeneration and neuroinflammation in amyotrophic lateral sclerosis. J. Neurol. 2020;267:1699–1708. doi: 10.1007/S00415-020-09761-Z. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

