The structure and function of mitofusin 2 and its role in cardiovascular disease through mediating mitochondria-associated endoplasmic reticulum membranes
- PMID: 40520935
- PMCID: PMC12163069
- DOI: 10.3389/fcvm.2025.1535401
The structure and function of mitofusin 2 and its role in cardiovascular disease through mediating mitochondria-associated endoplasmic reticulum membranes
Abstract
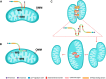
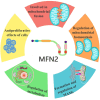
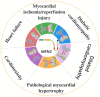
Cardiovascular disease (CVD) remains a leading cause of death globally, posing a major public health challenge. Due to the complexity of CVD's etiology, understanding its pathogenesis has been a significant challenge and research focus. In recent years, the communication between organelles has gained increasing attention, with mitochondria-associated endoplasmic reticulum (ER) membranes (MAMs) emerging as a key structural component that facilitates dialogue between the mitochondria and the ER. Numerous studies have highlighted that proteins located in MAMs may play a role in the development of CVD. Among these, mitofusin 2 (MFN2), a protein found on the outer mitochondrial and ER membranes, has garnered particular interest due to its widespread presence in MAMs. This review aims to sort out current research on MFN2, focusing on its potential involvement in myocardial protection through its mediation of MAMs. We discuss how MFN2-mediated MAMs may contribute to the protection against various CVDs, including myocardial ischemia/reperfusion injury, diabetic cardiomyopathy, dilated cardiomyopathy, pathological myocardial hypertrophy, cardiotoxicity, and heart failure. However, given the functional diversity of MFN2, the current body of research remains controversial, and further studies are urgently needed to clarify its precise mechanisms of action.
Keywords: cardioprotection; cardiovascular disease; endoplasmic reticulum; mitochondria; mitochondria-associated endoplasmic reticulum membranes; mitofusin 2.
© 2025 Lv, Chen, Li, Liao, Fang, Cheng and Peng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources

