Nonradical Reactions of p-Benzyne Diradicals
- PMID: 40521681
- PMCID: PMC12210243
- DOI: 10.1021/acs.joc.5c00517
Nonradical Reactions of p-Benzyne Diradicals
Abstract
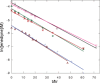
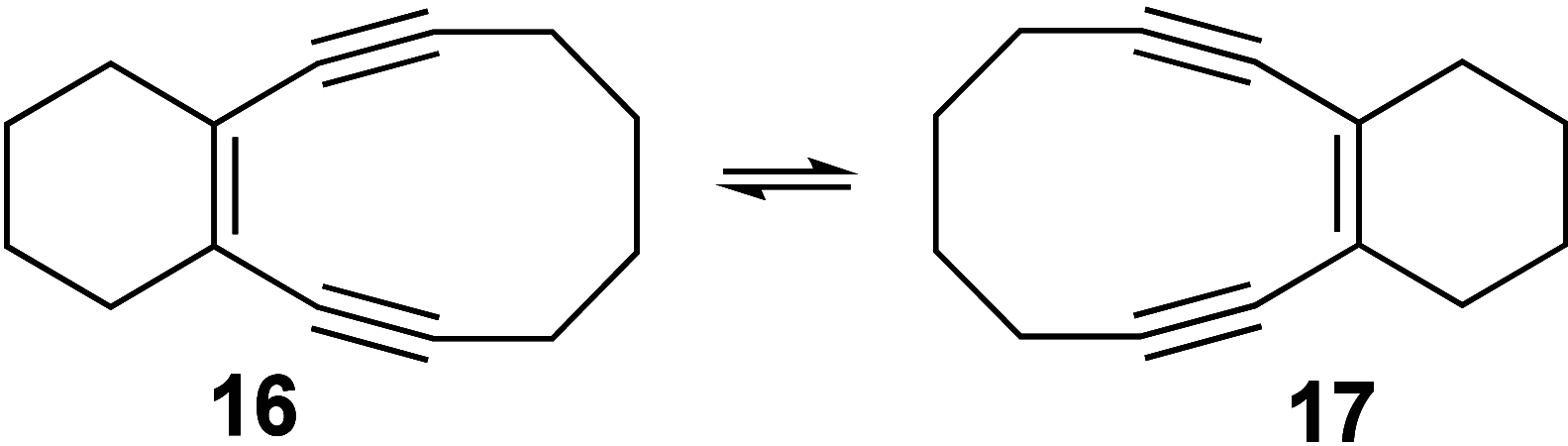
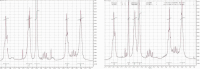
Enediynes undergo electrocyclization to p-benzyne diradicals. Although such diradicals ordinarily react by radical pathways, they can also be trapped by nucleophilic addition, a pathway supported by computations. The product of that addition is a "naked" aryl anion that is unselective toward protonation. A cyclohexeno-substituted enediyne was synthesized and found to be more amenable to further derivatization and mechanistic study. Moreover, it was shown to undergo automerization, rearrangement to a degenerate form of the original enediyne.
Figures
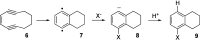



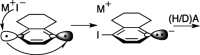
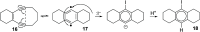
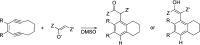



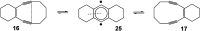
References
-
- Abe M.. Diradicals. Chem. Rev. 2013;113:7011–7088. doi: 10.1021/cr400056a. - DOI - PubMed
- Wenk H. H., Winkler M., Sander W.. One Century of Aryne Chemistry. Angew. Chem., Int. Ed. 2003;42:502–528. doi: 10.1002/anie.200390151. - DOI - PubMed
- Clark A. E., Davidson E. R.. Model Studies of Hydrogen Atom Addition and Abstraction Processes Involving ortho-, meta-, and para-Benzyne. J. Am. Chem. Soc. 2001;123:10691–10698. doi: 10.1021/ja0159620. - DOI - PubMed
-
- Perrin C. L., Agranat I., Bagno A., Braslavsky S. E., Fernandes P. A., Gal J.-F., Lloyd-Jones G. C., Mayr H., Murdoch J. R., Nudelman N. S., Radom L., Rappoport Z., Ruasse M.-F., Siehl H.-U., Takeuchi Y., Tidwell T. T., Uggerud E., Williams I. H.. Glossary of Terms Used in Physical Organic Chemistry (IUPAC Recommendations 2021) Pure Appl. Chem. 2022;94:353–534. doi: 10.1515/pac-2018-1010. - DOI
Publication types
LinkOut - more resources
Full Text Sources

