Targeting SIX2 as a novel sensitization strategy of sorafenib treatment on advanced hepatocellular carcinoma through modulating METTL9-SLC7A11 axis
- PMID: 40523929
- PMCID: PMC12170904
- DOI: 10.1038/s41698-025-01004-6
Targeting SIX2 as a novel sensitization strategy of sorafenib treatment on advanced hepatocellular carcinoma through modulating METTL9-SLC7A11 axis
Abstract
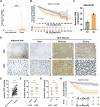
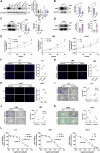
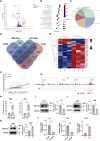
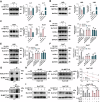
Sorafenib is the main treatment for advanced hepatocellular carcinoma (HCC), but drug resistance limits its effectiveness. Evidence increasingly indicates that, in addition to targeting tyrosine kinases, sorafenib also induces ferroptosis. However, current studies have not fully clarified the relationship between ferroptosis and sorafenib treatment sensitivity. Our bioinformatics analysis identified that SIX Homeobox 2 (SIX2), known for maintaining cellular stemness via the Wnt signaling pathway, was significantly upregulated in sorafenib-resistant tissues. Overexpression and knockdown experiments revealed that altering SIX2 expression affected HCC cell sensitivity to sorafenib and involved the ferroptosis pathway, suggesting a regulatory role for SIX2 in ferroptosis. RNA sequencing and CUT&Tag analysis showed that SIX2 directly regulated methyltransferase 9 (METTL9) expression. Co-immunoprecipitation (Co-IP) assays confirmed that METTL9 bound to SLC7A11, enhancing its stability and reducing degradation, thus regulating ferroptosis. Importantly, the role of SIX2 in ferroptosis operated independently of the classical glutathione peroxidase 4 (GPX4) pathway. In vitro studies further supported these findings, demonstrating that SIX2 knockdown increased sorafenib-induced ferroptosis in HCC, while METTL9 overexpression largely counteracted the effects of SIX2 knockdown. In mouse models, overexpression of SIX2 increased tumor resistance to sorafenib. Our findings suggest that modulating the ferroptosis pathway through SIX2 could enhance sorafenib sensitivity. This study provides the first evidence that SIX2 influences ferroptosis via the METTL9-SLC7A11 axis, thereby sensitizing HCC cells to sorafenib. Reducing SIX2 expression could thus represent a promising strategy to improve the efficacy of sorafenib in advanced HCC.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




References
-
- Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin.68, 394–424 (2018). - PubMed
-
- Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin.70, 7–30 (2020). - PubMed
-
- Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Prim.2, 16018 (2016). - PubMed
-
- Smith, R. A. et al. Cancer screening in the United States, 2019: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J. Clin.69, 184–210 (2019). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

