Unmet needs of patients with COPD in Germany: a retrospective, cross-sectional study
- PMID: 40524930
- PMCID: PMC12168169
- DOI: 10.1183/23120541.00976-2024
Unmet needs of patients with COPD in Germany: a retrospective, cross-sectional study
Abstract
Background: Earlier diagnosis and treatment of COPD, particularly preventing exacerbations, are key to slowing disease progression and reducing mortality. This study focused on the identification of patients in Germany with unstable COPD due to suboptimal treatments.
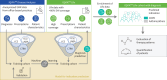
Methods: The IQVIA™ LRx database, capturing 80% of Statutory Health Insurance prescriptions was used to identify patients with COPD using a machine-learning model. Patients with unstable COPD were identified through high prescriptions of oral corticosteroid (OCS) and/or rescue inhalers between April 2022 and March 2023.
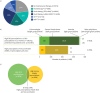
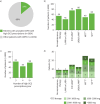
Results: The machine-learning model identified around 2.6 million treated patients with COPD, with 77% precision. The mean age was 71 years, 48% were female and 86% were aged ≥60 years. About 14% patients (n=363k) exhibited unstable COPD due to high OCS prescriptions, while 10% patients (n=256k) had high rescue inhaler prescriptions. Among those with high OCS and high rescue inhaler prescriptions, respectively, 43% and 38% were on dual therapy, 17% and 21% were on single inhaler triple therapy, 14% and 16% were on multiple inhaler triple therapy, 11% and 9% were on monotherapy and 15% and 17% had no maintenance therapy.
Conclusions: A substantial number of unstable COPD patients were either on suboptimal maintenance therapy (monotherapy or inhaled corticosteroid-based dual therapy) or not receiving any maintenance therapy. The study highlights a substantial need in Germany for improved maintenance therapy, which could reduce disease burden, improve disease stability and reduce reliance on OCS and rescue therapies, thereby minimising side effects.
Copyright ©The authors 2025.
Conflict of interest statement
Conflict of interest: F.J.F. Herth received grants or contracts from the German Ministry of Education and Science (BMBF), AstraZeneca, Boehringer Ingelheim, Chiesi, CSL Behring, GSK, Grifols and Novartis; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from AstraZeneca, CSL Behring, Chiesi, GSK, Insmed, Menarini, Novartis, Roche, Sanofi, Olympus and Pulmonx; and is an associate editor of this journal. Conflict of interest: C.F. Vogelmeier received grants or contracts from BMBF, AstraZeneca, Boehringer Ingelheim, Chiesi, CSL Behring, GSK, Grifols and Novartis; consulting fees from Aerogen, AstraZeneca, Boehringer Ingelheim, CSL Behring, Chiesi, GSK, Insmed, Menarini, Novartis, Nuvaira and Sanofi; and payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from Aerogen, AstraZeneca, Boehringer Ingelheim, CSL Behring, Chiesi, GSK, Insmed, Menarini, Novartis, Roche and Sanofi. Conflict of interest: F.C. Trudzinski received all support for the present manuscript from GSK; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from Novartis AG, GSK, Chiesi, Boehringer Ingelheim GmbH, Grifols, CSL Behring, Streamed Up, RG Gesellschaft für Information and Organisation GmbH and AstraZeneca; support for attending meetings and/or travel from Grifols and CSL Behring. Conflict of interest: H. Watz received grants or contracts from AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, Novartis, EpiEndo, Bristol Myers Squibb and Roche; consulting fees, payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events, support for attending meetings and/or travel from AstraZeneca, Boehringer Ingelheim, Chiesi and GSK; and participated on a data safety monitoring or advisory board for AstraZeneca, Boehringer Ingelheim, Chiesi and GSK. Conflict of interest: D. Skowasch received all support for the present manuscript from GSK; grants or contracts from Boehringer Ingelheim, BMBF and DFG; consulting fees from GSK, AstraZeneca, Sanofi, Janssen and MSD; and payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from GSK, AstraZeneca, Sanofi, Janssen, MSD, Chiesi and Boehringer Ingelheim. Conflict of interest: K-M. Beeh received consulting fees from GSK, Clario, Berlin Chemie, Chiesi, Sanofi and Bosch Healthcare; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from GSK, Berlin Chemie, Chiesi, Sanofi, Novartis, Orion Pharma and Hormosan; and participated on a data safety monitoring or advisory board for Clario and Chiesi. Conflict of interest: C. Compton, J. Claussen and T. Mohan hold financial equities in GSK and are employees of GSK. Conflict of interest: H. Richter is an employee of IQVIA GmbH & Co OHG; IQVIA were contracted and paid by GSK to make the data available and conduct the analyses. Conflict of interest: S. Bartel is an employee of GSK, holds financial equities in GSK and has received all support for the present manuscript from GSK GmbH & Co KG.
Figures

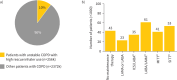

References
-
- Akmatov MK, Steffen A, Holstiege J, Bätzing J. Chronic obstructive pulmonary disease (COPD) in ambulatory care in Germany—Temporal trends and small-area variations. Berlin: Central Institute for Statutory Health Insurance in Germany (Zi); 2019. 15 August 2019. Report No. 19/06.
-
- Global Initiative for Chronic Obstructive Lung Disease . Global Strategy for the Diagnosis, Management, and Prevention of COPD: 2024 Report. Date last accessed: 6 May 2024. Date last updated: 31 December 2023. https://goldcopd.org/wp-content/uploads/2024/02/GOLD-2024_v1.2-11Jan24_W...
-
- Wedzicha JA, Mackay AJ, SIngh R. COPD exacerbations: impact and prevention. Breathe 2013; 9: 434–440. doi: 10.1183/20734735.002913 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
