Effects of corticothalamic feedback depend on visual responsiveness and stimulus type
- PMID: 40524960
- PMCID: PMC12167816
- DOI: 10.1016/j.isci.2025.112481
Effects of corticothalamic feedback depend on visual responsiveness and stimulus type
Abstract
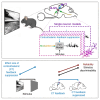
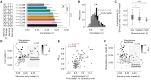
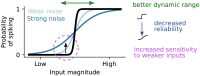
In the dorsolateral geniculate nucleus (dLGN) of the thalamus, stimulus-driven signals combine with modulatory inputs such as corticothalamic (CT) feedback and behavioral state, but their impact in shaping dLGN activity is debated. We recorded extracellular responses in the dLGN of mice viewing a movie stimulus, while photosuppressing CT feedback and tracking locomotion and pupil size. Using generalized linear models fit to single neuron responses, we found that including CT feedback and behavioral state improved model predictions, especially for a subpopulation of neurons poorly responsive to the movie. Intriguingly, the impact of CT feedback was stronger without a patterned visual stimulus. Finally, for neurons sensitive to CT feedback, visual stimuli could be more easily discriminated when CT feedback was suppressed. Together, these results show that the effects of modulatory inputs in dLGN depend on visual responsiveness and stimulus type, with CT feedback affecting sensitivity and reliability, potentially to tune the thalamic relay.
Keywords: Neuroscience; Sensory neuroscience.
© 2025 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Robust effects of corticothalamic feedback and behavioral state on movie responses in mouse dLGN.Elife. 2022 Mar 22;11:e70469. doi: 10.7554/eLife.70469. Elife. 2022. PMID: 35315775 Free PMC article.
-
Brain State-Dependent Modulation of Thalamic Visual Processing by Cortico-Thalamic Feedback.J Neurosci. 2023 Mar 1;43(9):1540-1554. doi: 10.1523/JNEUROSCI.2124-21.2022. Epub 2023 Jan 18. J Neurosci. 2023. PMID: 36653192 Free PMC article.
-
Optogenetic stimulation of the corticothalamic pathway affects relay cells and GABAergic neurons differently in the mouse visual thalamus.PLoS One. 2012;7(9):e45717. doi: 10.1371/journal.pone.0045717. Epub 2012 Sep 20. PLoS One. 2012. PMID: 23029198 Free PMC article.
-
The influence of the corticothalamic projection on responses in thalamus and cortex.Philos Trans R Soc Lond B Biol Sci. 2002 Dec 29;357(1428):1823-34. doi: 10.1098/rstb.2002.1159. Philos Trans R Soc Lond B Biol Sci. 2002. PMID: 12626015 Free PMC article. Review.
-
Corticothalamic interactions in the transfer of visual information.Philos Trans R Soc Lond B Biol Sci. 2002 Dec 29;357(1428):1739-52. doi: 10.1098/rstb.2002.1170. Philos Trans R Soc Lond B Biol Sci. 2002. PMID: 12626008 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

