Prevalence and Whole Genome Sequence Analysis of Mycoplasma bovis Isolates From Bulk Tank Milk of Dairy Farms in Tennessee, USA
- PMID: 40525812
- PMCID: PMC12172111
- DOI: 10.1111/jvim.70164
Prevalence and Whole Genome Sequence Analysis of Mycoplasma bovis Isolates From Bulk Tank Milk of Dairy Farms in Tennessee, USA
Abstract
Background: Mycoplasma bovis mastitis is an important disease of dairy cows that causes substantial economic losses. However, its prevalence in different states in the United States (US), including Tennessee, is not well known. Furthermore, recent studies showed a high prevalence of bovine hemotropic mycoplasmas in US dairy farms.
Objectives: Determine the prevalence of M. bovis in bulk tank milk (BTM) of dairy farms in Tennessee and evaluate the genetic diversity, virulence factors, and antimicrobial resistance genes of the identified isolates. In addition, the prevalence of Mycoplasma wenyonii and Candidatus Mycoplasma haemobos in the bulk tank milk was determined.
Methods: Seventy-five BTM samples were collected from 59 dairy farms. Of the 59 farms, 56 are in Tennessee and the remaining 3 farms are in the neighboring states, Georgia (n = 2) and Alabama (n = 1). Milk samples were tested using bacterial culture, PCR, and qPCR. M. bovis isolates were genetically characterized by pangenome analysis.
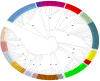
Results: Of the 56 farms, 3 (5.3%) were positive for M. bovis by bacterial culture and 43 (76.7%) were positive by PCR. Pangenome analysis showed clustering of current isolates with mastitis isolates from the US, Israel, and Europe. Of 75 BTM samples tested by qPCR, 42 (56%) and 51 (68%) were positive for M. wenyonii and C. M. haemobos, respectively.
Conclusions: M. bovis intramammary infection is prevalent in Tennessee dairy farms.
Keywords: M. bovis; Mycoplasma wenyonii; Candidatus Mycoplasma haemobos; mastitis; virulence factors.
© 2025 The Author(s). Journal of Veterinary Internal Medicine published by Wiley Periodicals LLC on behalf of American College of Veterinary Internal Medicine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Presence of Mycoplasma bovis in Bulk Tank Milk and Associated Risk Factor Analysis in Serbian Dairy Farms.Pathogens. 2024 Apr 6;13(4):302. doi: 10.3390/pathogens13040302. Pathogens. 2024. PMID: 38668257 Free PMC article.
-
Molecular characteristics and antibiotic susceptibility profiles of Mycoplasma bovis associated with mastitis on dairy farms in China.Prev Vet Med. 2020 Sep;182:105106. doi: 10.1016/j.prevetmed.2020.105106. Epub 2020 Aug 15. Prev Vet Med. 2020. PMID: 32810702
-
Antimicrobial resistance and molecular epidemiology of Klebsiella pneumoniae isolated from bovine mastitis in seven provinces in China.BMC Microbiol. 2025 Jul 3;25(1):407. doi: 10.1186/s12866-025-04147-5. BMC Microbiol. 2025. PMID: 40604415 Free PMC article.
-
Biofilm Formation in Dairy: A Food Safety Concern-Insights into the prevalence of Pseudomonadota and yeasts on milking system surface biofilms.J Dairy Sci. 2025 Aug;108(8):8141-8156. doi: 10.3168/jds.2024-26016. Epub 2025 Jul 16. J Dairy Sci. 2025. PMID: 40675481 Review.
-
Toxigenic Corynebacterium ulcerans in raw milk of a cow with acute mastitis: case report and historical review on milk-transmitted diphtheria.Infection. 2025 Jun;53(3):767-774. doi: 10.1007/s15010-025-02477-0. Epub 2025 Feb 20. Infection. 2025. PMID: 39976805 Review.
References
-
- Jordan A., Sadler R. J., Sawford K., Andel M., Ward M., and Cowled B. D., “ <styled-content style="fixed-case"> Mycoplasma bovis </styled-content> Outbreak in New Zealand Cattle: An Assessment of Transmission Trends Using Surveillance Data,” Transboundary and Emerging Diseases 68 (2021): 3381–3395. - PubMed
-
- Maunsell F. P., Woolums A. R., Francoz D., et al., “ Mycoplasma bovis Infections in Cattle,” Journal of Veterinary Internal Medicine 25 (2011): 772–783. - PubMed
-
- Nicholas R. A. and Ayling R. D., “ Mycoplasma bovis: Disease, Diagnosis, and Control,” Research in Veterinary Science 74 (2003): 105–112. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

