CuATSM Enhances Wound Repair Without Scarring via Hippo/YAP Signalling Pathway to Reduce Ferroptosis and Macrophage Polarisation
- PMID: 40525915
- PMCID: PMC12172343
- DOI: 10.1111/jcmm.70590
CuATSM Enhances Wound Repair Without Scarring via Hippo/YAP Signalling Pathway to Reduce Ferroptosis and Macrophage Polarisation
Abstract
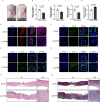
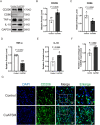
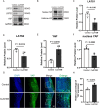
Skin wound healing is a complex biological process involving haemostasis, inflammation, proliferation/repair and remodelling. However, skin scarring, as one of the important stages in the healing process, can adversely affect the structure and function of related organs. Currently, effective treatments to address such scars remain insufficient. In this study, we established a full-thickness skin excision wound model using male ICR mice, which were randomly divided into a Control group and a CuATSM group. The CuATSM group received CuATSM (30 mg/kg) via gavage, with daily treatments continuing throughout the observation period. The Control group received an equivalent volume of 0.9% sodium chloride solution. Wound healing progression was evaluated through macroscopic photography, histological analyses, Western blotting and quantification of relevant biochemical markers at different healing stages. Our study reveals that CuATSM not only promotes rapid skin wound healing but also reduces scar formation in the late healing phase. Furthermore, our findings suggest that this effect is mediated through the ferroptosis-induced Hippo/YAP signalling pathway and macrophage polarisation. These findings highlight CuATSM as a promising therapeutic candidate for achieving scarless wound repair in clinical applications.
Keywords: CuATSM; ferroptosis; hippo/YAP pathway; scarless; wound healing.
© 2025 The Author(s). Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Angiotensin II depends on hippo/YAP signaling to reprogram angiogenesis and promote liver fibrosis.Cell Signal. 2024 Nov;123:111355. doi: 10.1016/j.cellsig.2024.111355. Epub 2024 Aug 22. Cell Signal. 2024. PMID: 39173854
-
Activation of Hippo/YAP signaling pathway exacerbates vascular remodeling and aggravates hypertension by upregulating Foxm1.J Mol Histol. 2025 May 19;56(3):158. doi: 10.1007/s10735-025-10443-1. J Mol Histol. 2025. PMID: 40388085
-
Hippo pathway-mediated YAP1/TAZ inhibition is essential for proper pancreatic endocrine specification and differentiation.Elife. 2024 Jul 25;13:e84532. doi: 10.7554/eLife.84532. Elife. 2024. PMID: 39051998 Free PMC article.
-
Assessing the comparative effects of interventions in COPD: a tutorial on network meta-analysis for clinicians.Respir Res. 2024 Dec 21;25(1):438. doi: 10.1186/s12931-024-03056-x. Respir Res. 2024. PMID: 39709425 Free PMC article. Review.
-
[Research progress on the regulation of Hippo -YAP signaling pathway in osteoarthritis].Zhongguo Gu Shang. 2025 Jul 25;38(7):759-64. doi: 10.12200/j.issn.1003-0034.20230522. Zhongguo Gu Shang. 2025. PMID: 40759556 Review. Chinese.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

