The Ascent of Supramolecular Polymers and Gels in Asymmetric Catalysis
- PMID: 40528528
- PMCID: PMC12284627
- DOI: 10.1002/chem.202501446
The Ascent of Supramolecular Polymers and Gels in Asymmetric Catalysis
Abstract
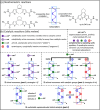
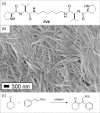
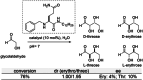
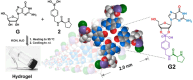
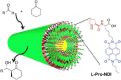
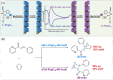
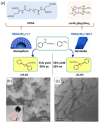
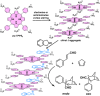
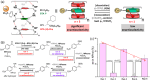
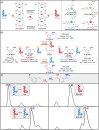
Supramolecular polymers (SPs) and gels, formed by the spontaneous assembly of small molecules through various types of noncovalent interactions, are attractive materials for many applications. Their modularity also offers many opportunities in asymmetric catalysis that have been tackled in the last two decades and more intensively in the last one. In this review, strategies adopted to develop efficient asymmetric catalysts supported on SPs and gels are first categorized according to the chiral or achiral nature of the monomers used for their construction and second to their ability to be commuted into different states. Catalytic SPs have been described for which enantioselectivity stems mostly from the molecular chirality located next to the reactive group, or at opposite ends of the spectrum, exclusively from the chiral environment provided by the supramolecular helices. New paradigms revealed by these systems include (i) the organization of catalytic sites at the periphery of modular and well-structured 1D assemblies, (ii) the possibility to conduct asymmetric reactions with a sub-catalytic amount of chiral inducers and even in the absence of chiral monomers, and (iii) the development of a new class of switchable asymmetric catalysts.
Keywords: asymmetric catalysis; chirality; supramolecular gels; supramolecular polymers; synthesis.
© 2025 The Author(s). Chemistry – A European Journal published by Wiley‐VCH GmbH.
Conflict of interest statement
There are no conflicts to declare.
Figures












Similar articles
-
The Role of Anions in Guanidinium-Catalyzed Chiral Cation Ion Pair Catalysis.Acc Chem Res. 2025 Jul 15;58(14):2269-2281. doi: 10.1021/acs.accounts.5c00283. Epub 2025 Jun 30. Acc Chem Res. 2025. PMID: 40587427
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
Induced-Fit Chiral N-Heterocyclic Carbene Ligands for Asymmetric Catalysis.Acc Chem Res. 2025 Jul 1;58(13):2157-2177. doi: 10.1021/acs.accounts.5c00304. Epub 2025 Jun 19. Acc Chem Res. 2025. PMID: 40536020
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
References
-
- De Greef T. F. A., Smulders M. M. J., Wolffs M., Schenning A. P. H. J., Sijbesma R. P., Meijer E. W., Chem. Rev. 2009, 109, 5687. - PubMed
-
- Hart L. R., Harries J. L., Greenland B. W., Colquhoun H. M., Hayes W., Polym. Chem. 2013, 4, 4860.
-
- Petkau‐Milroy K., Sonntag M. H., Brunsveld L., Chem. Eur. J. 2013, 19, 10786. - PubMed
-
- Dong R., Zhou Y., Huang X., Zhu X., Lu Y., Shen J., Adv. Mater. 2015, 27, 498. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

