MicroRNA-29a-5p attenuates hemorrhagic transformation and improves outcomes after mechanical reperfusion for acute ischemic stroke
- PMID: 40529227
- PMCID: PMC12173523
- DOI: 10.1016/j.ncrna.2025.05.016
MicroRNA-29a-5p attenuates hemorrhagic transformation and improves outcomes after mechanical reperfusion for acute ischemic stroke
Abstract
Background: Hemorrhage transformation (HT) following endovascular reperfusion treatment is associated with worse clinical outcomes in acute ischemic stroke patients. MicroRNA (miR) modulates several aspects of cerebral ischemia-reperfusion injury, including blood-brain barrier (BBB) integrity, inflammation, oxidative stress, and apoptosis, significantly impacting cerebral recovery and function. This study investigated the role of astrocytic miR-29a-5p in HT in the transient middle cerebral artery occlusion (MCAO) model and oxygen-glucose deprivation reoxygenation (OGD/R) model of astrocytes.
Methods: MiR-29a-5p expression in the OGD/R astrocyte model was assessed. The astrocyte injury, the expression of A1 and A2 phenotypes of reactive astrocytes, and the regulation of miR-29a-5p target genes were evaluated after the miR-29a-5p intervention. A mechanical reperfusion-induced HT model was established in hyperglycemic rats using 5-h MCAO following reperfusion at 6 h. MiR-29a-5p agomir was administered intravenously before reperfusion. Infarct volume, HT, BBB damage, neurological score, the expression of miR-29a-5p, and its target genes were evaluated.
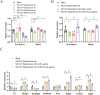
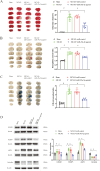
Results: MiR-29a-5p expression decreased in OGD/R-treated astrocytes and the peri-infarction tissue and blood of the MCAO model. Elevating miR-29a-5p levels reduced astrocyte injury, suppressed neurotoxic A1 astrocyte markers (C3, Fkbp5, and Serping1), while enhanced neuroprotective A2 astrocyte markers (S100a10 and Emp1) in the OGD/R and MCAO models. Intravenous administration of miR-29a-5p agomir increased the expression of miR-29a-5p and reduced infarct volume, reperfusion-induced HT, and BBB breakdown after ischemia, improving neurological outcomes in the MCAO model. Overexpression of miR-29a-5p effectively suppressed the expression of its direct target genes, glycogen synthase kinase 3 beta and aquaporin 4 in the OGD/R and MCAO models.
Conclusions: MiR-29a-5p alleviates astrocyte injury and regulates A1 and A2 astrocyte markers, glycogen synthase kinase 3 beta, and aquaporin 4 in astrocytes subjected to ischemia-reperfusion injury. Astrocytic miR-29a-5p may be a protective target for reducing HT and improving outcomes following mechanical reperfusion in acute ischemic stroke.
Keywords: Acute ischemic stroke; Astrocyte; Cerebral ischemia-reperfusion injury; Hemorrhagic transformation; MicroRNA; Oxygen-glucose deprivation reoxygenation.
© 2025 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


Similar articles
-
TAT and RVG Co-modified MSC-derived Exosomes-mediated Delivery of microRNA-15b-5p Inhibitor Alleviate Cerebral Ischemia and Reperfusion-induced Neuronal Apoptosis by Promoting HTR2C-ERK Signaling.Mol Neurobiol. 2025 Jul 25. doi: 10.1007/s12035-025-05249-x. Online ahead of print. Mol Neurobiol. 2025. PMID: 40711710
-
Knockdown of RUNX2 Attenuated A1 Astrocyte Overactivation, Brain Injury, and Cerebral Edema During Ischemic Stroke.Neuromolecular Med. 2025 Jun 27;27(1):48. doi: 10.1007/s12017-025-08868-8. Neuromolecular Med. 2025. PMID: 40576866
-
Naloxone modulates the miR-30a-5p/ follistatin-like 1 axis to attenuate neurological deficits and hippocampal neuronal injury in ischemic stroke.J Physiol Pharmacol. 2025 Jun;76(3). doi: 10.26402/jpp.2025.3.04. Epub 2025 Jul 16. J Physiol Pharmacol. 2025. PMID: 40698786
-
A systematic review of the research progress of non-coding RNA in neuroinflammation and immune regulation in cerebral infarction/ischemia-reperfusion injury.Front Immunol. 2022 Oct 7;13:930171. doi: 10.3389/fimmu.2022.930171. eCollection 2022. Front Immunol. 2022. PMID: 36275741 Free PMC article.
-
Matrix metalloproteinase‑9 in hemorrhagic transformation after acute ischemic stroke (Review).Mol Med Rep. 2025 Aug;32(2):225. doi: 10.3892/mmr.2025.13590. Epub 2025 Jun 6. Mol Med Rep. 2025. PMID: 40476578 Free PMC article. Review.
References
-
- Majoie C.B., Cavalcante F., Gralla J., Yang P., Kaesmacher J., Treurniet K.M., et al. Value of intravenous thrombolysis in endovascular treatment for large-vessel anterior circulation stroke: individual participant data meta-analysis of six randomised trials. Lancet. 2023;402:965–974. doi: 10.1016/S0140-6736(23)01142-X. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

