Delivery strategies for RNA-targeting therapeutic nucleic acids and RNA-based vaccines against respiratory RNA viruses: IAV, SARS-CoV-2, RSV
- PMID: 40529300
- PMCID: PMC12173128
- DOI: 10.1016/j.omtn.2025.102572
Delivery strategies for RNA-targeting therapeutic nucleic acids and RNA-based vaccines against respiratory RNA viruses: IAV, SARS-CoV-2, RSV
Abstract
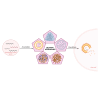
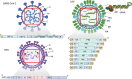
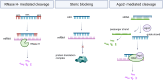
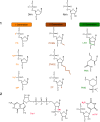
Therapeutic nucleic acids, including small interfering RNA (siRNA), and antisense oligonucleotides (ASOs), targeting RNA viruses such as influenza A virus (IAV), severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and respiratory syncytial virus (RSV), play a crucial role in contemporary medicine. The primary goal of short oligonucleotide-based antivirals is to precisely inhibit viral mechanisms by interacting with viral RNA, thereby opening new avenues for infection treatment. RNA recently was also used to invent mRNA vaccine for different illness prevention. Therapeutic nucleic acids and mRNA vaccine attracted considerable attention during the COVID-19 pandemic due to the pressing necessity to develop an effective strategy to address this global threat. In addition to the advancement of therapeutic nucleic acids aimed at targeting respiratory viruses, the effective delivery of these molecules to infected cells is of paramount importance. Similarly, mRNA vaccine's effectiveness also depends on effective delivery. This article offers a comprehensive summary and analysis of various delivery strategies, along with the challenges encountered in their development. Representative studies conducted in cellular models, model organisms, and human are presented for examination. Furthermore, the article explores future perspectives regarding the delivery of therapeutic nucleic acids and mRNA vaccines aimed at combating IAV, SARS-CoV-2, and RSV.
Keywords: ASO; MT: Delivery Strategies; RNA therapeutics; RSV; SARS-CoV-2; delivery methods; influenza A; mRNA vaccine; oligonucleotides.
© 2025 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Performance Evaluation of a Fully Automated Molecular Diagnostic System for Multiplex Detection of SARS-CoV-2, Influenza A/B Viruses, and Respiratory Syncytial Virus.Diagnostics (Basel). 2025 Jul 16;15(14):1791. doi: 10.3390/diagnostics15141791. Diagnostics (Basel). 2025. PMID: 40722545 Free PMC article.
-
Detection methods and dynamic characteristics of specific antibodies in patients with COVID-19: A review of the early literature.Heliyon. 2024 Jan 24;10(3):e24580. doi: 10.1016/j.heliyon.2024.e24580. eCollection 2024 Feb 15. Heliyon. 2024. Retraction in: Heliyon. 2025 Mar 25;11(9):e43262. doi: 10.1016/j.heliyon.2025.e43262. PMID: 38317938 Free PMC article. Retracted. Review.
-
Analysis of the SARS-CoV-2 inactivation mechanism using violet-blue light (405 nm).Appl Environ Microbiol. 2025 Jun 18;91(6):e0040325. doi: 10.1128/aem.00403-25. Epub 2025 May 14. Appl Environ Microbiol. 2025. PMID: 40366184 Free PMC article.
-
An intranasal ASO therapeutic targeting SARS-CoV-2.Nat Commun. 2022 Aug 3;13(1):4503. doi: 10.1038/s41467-022-32216-0. Nat Commun. 2022. PMID: 35922434 Free PMC article.
-
Assessing the comparative effects of interventions in COPD: a tutorial on network meta-analysis for clinicians.Respir Res. 2024 Dec 21;25(1):438. doi: 10.1186/s12931-024-03056-x. Respir Res. 2024. PMID: 39709425 Free PMC article. Review.
References
-
- Braasch D.A., Jensen S., Liu Y., Kaur K., Arar K., White M.A., Corey D.R. RNA interference in mammalian cells by chemically-modified RNA. Biochemistry. 2003;42:7967–7975. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

