Biomarker Discovery in Wilson's Disease-A Path Toward Improved Diagnosis and Management: A Comprehensive Review
- PMID: 40531364
- PMCID: PMC12511123
- DOI: 10.1007/s12035-025-05143-6
Biomarker Discovery in Wilson's Disease-A Path Toward Improved Diagnosis and Management: A Comprehensive Review
Abstract
Wilson's disease (WD) is a rare autosomal recessive disorder characterized by defective copper metabolism, which leads to hepatic and neurological damage. The clinical presentation of WD varies significantly, often resulting in delayed diagnosis and an increased risk of irreversible complications. Current diagnostic tools, including biochemical assays, imaging techniques, and genetic testing, lack sufficient specificity and sensitivity, highlighting the need for novel biomarkers for early diagnosis and treatment monitoring. This review explores emerging biomarkers for both hepatic and neurological manifestations of WD, including blood-based molecular markers such as cytokines, proteases, oxidative stress indicators, inflammasomes, and gut microbiota signatures. Recent studies have identified neurofilament light chain (NfL), pentraxin 3 (PTX3), caspase-3/XIAP, and NLRP3 inflammasome activation as promising indicators of neurological impairment. Additionally, markers like soluble CD163 (sCD163) and apoptosis antigen 1 (APO-1) show potential for assessing hepatic dysfunction. Metabolomic and proteomic analyses further suggest distinct molecular profiles associated with different WD subtypes, while microRNA-based biomarkers offer novel insights into disease progression. Identifying and validating these biomarkers could enhance early diagnosis, predict neurological deterioration, and optimize treatment strategies, ultimately improving patient outcomes. Further research is needed to integrate these biomarkers into clinical practice and establish standardized protocols for their use in WD management.
Keywords: Early diagnosis; Neurological impairment; Novel biomarkers; Treatment monitoring; Wilson’s disease.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics Approval and Consent to Participate: Not applicable. Patient Consent for Publication: Not applicable. Competing Interests: The authors declare no competing interests.
Figures
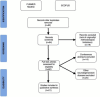

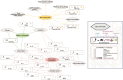

References
-
- El-Youssef M (2003) Wilson disease. Mayo Clin Proc 78:1126–1136. 10.4065/78.9.1126 - PubMed
-
- Deguti MM, Genschel J, Cancado ELR et al (2004) Wilson disease: novel mutations in the ATP7B gene and clinical correlation in Brazilian patients. Hum Mutat 23:398. 10.1002/humu.9227 - PubMed
-
- Huster D (2010) Wilson disease. Best Pract Res Clin Gastroenterol 24:531–539. 10.1016/j.bpg.2010.07.014 - PubMed
-
- A multidisciplinary approach to the diagnosis and management of Wilson disease: 2022 practice guidance on Wilson disease from the American Association for the Study of Liver Diseases. 10.1002/hep.32801. Accessed 4 Mar 2025 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

