Grain under pressure: Harnessing biochemical pathways to beat drought and heat in wheat
- PMID: 40532117
- PMCID: PMC12176370
- DOI: 10.1111/tpj.70253
Grain under pressure: Harnessing biochemical pathways to beat drought and heat in wheat
Abstract
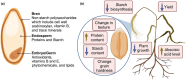
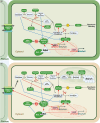
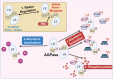
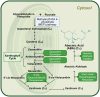
Erratic climate patterns represent a remarkable challenge to global food security, particularly affecting staple cereal crops of which wheat (Triticum aestivum) plays a critical role in annual agricultural production globally. It has been shown that over the last four decades, wheat cultivation has faced an escalating vulnerability to a variety of abiotic stresses, including heat and drought. These stressors not only decrease overall yield but also compromise grain quality, leading to reduced soluble starch content, higher protein content, altered grain texture, diminished end-use quality, and various other undesirable changes. With climate change projections indicating an intensification and higher frequency of heat and drought conditions in the future, urgent action is needed to develop resilient wheat varieties. Achieving this goal relies on a comprehensive understanding of the molecular responses to environmental shifts during successive stages of reproduction. Here we discuss three types of critical biochemical pathways responsible for sustaining starch biosynthesis in both source and sink tissues under adverse environmental conditions during grain development: (i) signaling network and cross-talk between ABA and SnRK pathways; (ii) transcriptional changes of the enzymes and signaling components; and (iii) inhibition of enzyme activity through temperature-induced misfolding. While summarizing the current knowledge, we also highlight critical factors contributing to the deterioration of grain quality and propose potential strategies for enhancing the resilience of starch biosynthesis in wheat grain.
Keywords: abiotic stress; biochemical pathways; drought; grain development; heat; starch biosynthesis; wheat.
© 2025 The Author(s). The Plant Journal published by Society for Experimental Biology and John Wiley & Sons Ltd.
Conflict of interest statement
The authors have not declared a conflict of interest.
Figures
References
-
- Ahmadi, A. & Baker, D.A. (1999) Effects of abscisic acid (ABA) on grain filling processes in wheat. Plant Growth Regulation, 28(3), 187–197. Available from: 10.1023/A:1006223925694 - DOI
-
- Al‐Sheikh Ahmed, S. , Zhang, J. , Ma, W. & Dell, B. (2018) Contributions of TaSUTs to grain weight in wheat under drought. Plant Molecular Biology, 98, 333–347. - PubMed
-
- Balla, K. , Rakszegi, M. , Li, Z. , Békés, F. , Bencze, S. & Veisz, O. (2011) Quality of winter wheat in relation to heat and drought shock after anthesis. Czech Journal of Food Sciences, 29(2), 117–128. Available from: 10.17221/227/2010-CJFS - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

