Evolving nutrition therapy in cystic fibrosis: Adapting to the CFTR modulator era
- PMID: 40533897
- PMCID: PMC12242106
- DOI: 10.1002/ncp.11332
Evolving nutrition therapy in cystic fibrosis: Adapting to the CFTR modulator era
Abstract
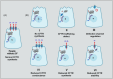
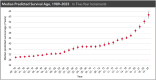
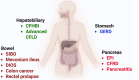
Cystic fibrosis transmembrane regulator (CFTR)-directed therapies, such as modulators, have transformed the medical management of people with CF, resulting in better lung function, weight, and body mass index in recent years. With improved nutrition status in people on CFTR modulators, the emphasis on a high-energy, high-fat diet (the legacy CF diet) is declining, with an increased focus on a healthy diet. The increased survival and median predicted age of people with CF have created a need for more attention to metabolic diseases, including hypertension, dyslipidemia, and cardiovascular diseases. The effects of modulators on extrapulmonary manifestations associated with CF, such as CF-related diabetes, CF hepatobiliary involvement, gastrointestinal tract disorders, and pancreatic manifestations, are currently unknown. Approximately 95% of people with CF qualify for treatment with a CFTR modulator. This review discusses the basics of CFTR gene mutations and changes in nutrition status related to treatment with CFTR modulators.
Keywords: CFTR gene mutations; CFTR modulators; CF‐related diabetes; CF‐related liver disease; aging; cardiometabolic risk; cystic fibrosis (CF); elexacaftor/tezacaftor/ivacaftor (ETI); exocrine pancreatic insufficiency; fat‐soluble vitamin; gastrointestinal manifestations; ivacaftor; legacy CF diet; nutritional therapy; obesity; overweight; vanzacaftor.
© 2025 The Author(s). Nutrition in Clinical Practice published by Wiley Periodicals LLC on behalf of American Society for Parenteral and Enteral Nutrition.
Conflict of interest statement
Senthilkumar Sankararaman serves as a consultant for Nestlé Health Science. Kay Vavrina serves on the speaker's bureaus of Alcresta Therapeutics, Abbvie, Nestlé Health Science, and DCI. The remaining authors declare no conflict of interests.
Figures
Similar articles
-
Corrector therapies (with or without potentiators) for people with cystic fibrosis with class II CFTR gene variants (most commonly F508del).Cochrane Database Syst Rev. 2023 Nov 20;11(11):CD010966. doi: 10.1002/14651858.CD010966.pub4. Cochrane Database Syst Rev. 2023. PMID: 37983082 Free PMC article.
-
Vanzacaftor-tezacaftor-deutivacaftor versus elexacaftor-tezacaftor-ivacaftor in individuals with cystic fibrosis aged 12 years and older (SKYLINE Trials VX20-121-102 and VX20-121-103): results from two randomised, active-controlled, phase 3 trials.Lancet Respir Med. 2025 Mar;13(3):256-271. doi: 10.1016/S2213-2600(24)00411-9. Epub 2025 Jan 2. Lancet Respir Med. 2025. PMID: 39756424 Free PMC article. Clinical Trial.
-
Pancreatic, nutritional and clinical outcomes in children 0-5 years with cystic fibrosis during the first 2 years of CFTR modulator therapy (PaNC): a multicentre prospective observational study protocol.BMJ Open. 2025 Jul 30;15(7):e097071. doi: 10.1136/bmjopen-2024-097071. BMJ Open. 2025. PMID: 40738633 Free PMC article.
-
Population Pharmacokinetics of Elexacaftor, Tezacaftor and Ivacaftor in a Real-World Cohort of Adults with Cystic Fibrosis.Clin Pharmacokinet. 2025 Jun;64(6):959-971. doi: 10.1007/s40262-025-01516-1. Epub 2025 May 22. Clin Pharmacokinet. 2025. PMID: 40405059
-
Pharmacokinetics of Ivacaftor, Tezacaftor, Elexacaftor, and Lumacaftor in Special Cystic Fibrosis Populations: A Systematic Review.Clin Pharmacokinet. 2025 Jul;64(7):999-1046. doi: 10.1007/s40262-025-01507-2. Epub 2025 May 21. Clin Pharmacokinet. 2025. PMID: 40399734 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

