Vagus Nerve Stimulation Inhibits DNA and RNA Methylation in a Rat Model of Pilocarpine-Induced Temporal Lobe Epilepsy
- PMID: 40534269
- PMCID: PMC12177201
- DOI: 10.1111/cns.70484
Vagus Nerve Stimulation Inhibits DNA and RNA Methylation in a Rat Model of Pilocarpine-Induced Temporal Lobe Epilepsy
Abstract
Introduction: Vagus nerve stimulation (VNS) represents a clinically approved neuromodulatory intervention for managing refractory epilepsy. VNS exhibits antiepileptogenic effects in animal models; the molecular and cellular mechanisms driving these effects have yet to be fully elucidated. Epigenetic alterations, including DNA and RNA methylation, are implicated in the pathogenesis of epilepsy. This study was designed to examine whether VNS inhibits DNA/RNA methylation in temporal lobe epilepsy.
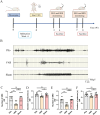
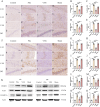
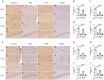
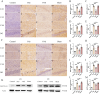
Methods: A rat model of temporal lobe epilepsy induced by intraperitoneal injection of lithium chloride-pilocarpine was used. Naïve control group, Pilo groups (8 and 12 weeks after pilocarpine respectively), VNS group (VNS stimulation for 4w and 8w respectively), and Sham group (VNS off control group, with VNS implanted but not turned on for 4w and 8w respectively). The rats were monitored by video-EEG. DNMT1 (DNA methyltransferase-1), DNMT3A (methyltransferase-3A), 5-hmC (5-hydroxymethyl-cytosine), 5-mC (5-methyl-cytosine), METTL3 (RNA methyltransferase like 3), METTL14 (RNA methyltransferase like 14) were quantified via immunohistochemistry and Western blot.
Results: Pilocarpine-induced epileptic animals were characterized by overexpression of 5-mC, DNMT1, DNMT3A, METTL3, and METTL14, and a decrease of 5-hmC. Compared to the Pilo group, VNS significantly inhibited the overexpression of 5-mC, DNMT1, DNMT3A, METTL3, and METTL14, reduced the downregulation of 5-hmC, and attenuated spontaneous recurrent seizures (SRSs).
Conclusions: We conclude that VNS reduces the number of SRSs in a rat model of epilepsy, concurrently reducing the expression of 5-mC, DNMT1, DNMT3A, METTL3, and METTL14, as well as the increase of 5-hmC. These findings suggest that epigenetic modifications, specifically the suppression of DNA and RNA methylation, could constitute a potential target of VNS-mediated antiepileptic effects.
Keywords: 5‐hmC; 5‐mC; METTL14; METTL3; epilepsy; vagus nerve stimulation.
© 2025 The Author(s). CNS Neuroscience & Therapeutics published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Tropisetron, an Antiemetic Drug, Exerts an Anti-Epileptic Effect Through the Activation of α7nAChRs in a Rat Model of Temporal Lobe Epilepsy.CNS Neurosci Ther. 2024 Oct;30(10):e70086. doi: 10.1111/cns.70086. CNS Neurosci Ther. 2024. PMID: 39445711 Free PMC article.
-
Baicalein Alleviates Lithium-Pilocarpine-Induced Status Epilepticus by Regulating DNMT1/GABRD Pathway in Rats.Organogenesis. 2025 Dec;21(1):2519607. doi: 10.1080/15476278.2025.2519607. Epub 2025 Jun 26. Organogenesis. 2025. PMID: 40569104 Free PMC article.
-
Alterations in DNA 5-hydroxymethylation patterns in the hippocampus of an experimental model of chronic epilepsy.Neurobiol Dis. 2024 Oct 1;200:106638. doi: 10.1016/j.nbd.2024.106638. Epub 2024 Aug 13. Neurobiol Dis. 2024. PMID: 39142613
-
Changes in sleep patterns after vagus nerve stimulation, deep brain stimulation or epilepsy surgery: Systematic review of the literature.Seizure. 2018 Mar;56:4-8. doi: 10.1016/j.seizure.2018.01.022. Epub 2018 Jan 31. Seizure. 2018. PMID: 29414594
-
Noninvasive Vagus Nerve Stimulation in Parkinson's Disease: A Systematic Review.Neuromodulation. 2025 Jun;28(4):641-651. doi: 10.1016/j.neurom.2025.01.012. Epub 2025 Mar 6. Neuromodulation. 2025. PMID: 40057881
References
-
- J. Engel, Jr. , Wiebe S., French J., et al., “Practice Parameter: Temporal Lobe and Localized Neocortical Resections for Epilepsy: Report of the Quality Standards Subcommittee of the American Academy of Neurology, in Association With the American Epilepsy Society and the American Association of Neurological Surgeons,” Neurology 60 (2003): 538–547, 10.1212/01.wnl.0000055086.35806.2d. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

